About The Report
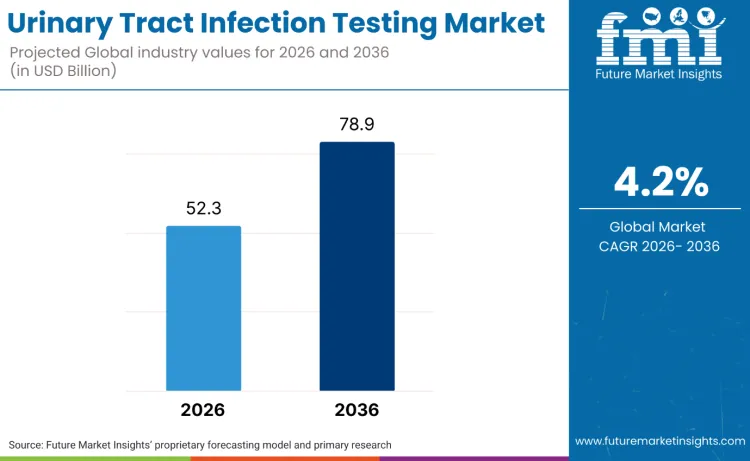
The global urinary tract infection testing market is forecasted to reach USD 52.3 billion in 2026 and expand to USD 78.9 billion by 2036, advancing at a CAGR of 4.2%. FMI states that this growth trajectory is being driven by increasing UTI prevalence across diverse demographics, particularly among women, the elderly, and hospital patients. Rising concerns over antibiotic resistance and the growing need for rapid, accurate diagnostics are transforming the way UTIs are detected and treated, fueling demand for advanced testing solutions.
Two key trends are accelerating market expansion. First, the increasing shift toward point-of-care (POC) diagnostics is reducing the reliance on traditional lab-based methods and enabling faster, more convenient testing in both clinical and home settings. Second, innovations in molecular diagnostics and the integration of artificial intelligence (AI) in testing platforms are improving accuracy, allowing for more personalized treatment regimens. These developments are making it possible to detect UTI pathogens with greater specificity, even in cases where symptoms are atypical.
The value proposition of UTI testing is evolving from traditional culture-based methods toward rapid, multiplexed diagnostic technologies. Urine dipsticks and PCR-based tests are gaining traction due to their ease of use, affordability, and ability to deliver results within hours rather than days. Additionally, the integration of digital health tools and mobile applications is enhancing post-testing care by enabling real-time monitoring of treatment progress.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 52.3 billion |
| Market Forecast Value (2036) | USD 78.9 billion |
| Forecast CAGR (2026-2036) | 4.2% |
Source: FMI’s proprietary forecasting model and primary research
Based on Future Market Insights’ analysis, pre‑2026, urinary tract infection (UTI) testing is largely centralized and culture‑dependent, requiring clinical visits and multi‑day lab turnaround. Healthcare systems relied on symptomatic assessment followed by empirical antibiotic prescribing before confirmatory results. This paradigm limited timely diagnosis and contributed to antimicrobial overuse.
In 2026, advances in point‑of‑care diagnostics and regulatory alignment propelled rapid molecular and immunoassay tests into frontline care. Standards for analytical accuracy and clinical validation replaced ambiguous performance claims. As a result, UTI tests transitioned from retrospective culture reporting to real‑time pathogen and resistance marker detection, improving targeted therapy decisions within hours rather than days.
Consumer engagement has changed significantly. Traditional UTI diagnostics focused on clinician‑driven workflows with little patient involvement. From 2026-2036, connected digital health tools and home‑use platforms empower patients with guided sample collection, symptom tracking, and results via secure apps. Integration with telehealth expanded access, enabling immediate clinician consultation and personalized treatment recommendations.
The development and deployment model also evolved. The previous decade’s reliance on large batch production and centralized labs could not keep pace with demand for rapid, decentralized diagnostics. AI‑assisted assay optimization, modular manufacturing, and flexible reagent production now support agile roll‑outs of tests tailored to regional pathogen profiles.
Urinary tract infection testing plays a critical role in delivering accurate diagnosis, rapid detection, and treatment guidance across clinical laboratories, point-of-care settings, and home testing applications. Adoption is influenced by accuracy requirements, cost considerations, turnaround time specifications, and clinical performance criteria. Segmentation by test type, technology, and end-user highlights how healthcare providers select specific testing solutions to meet diagnostic standards, cost targets, and clinical preferences across diverse healthcare settings.
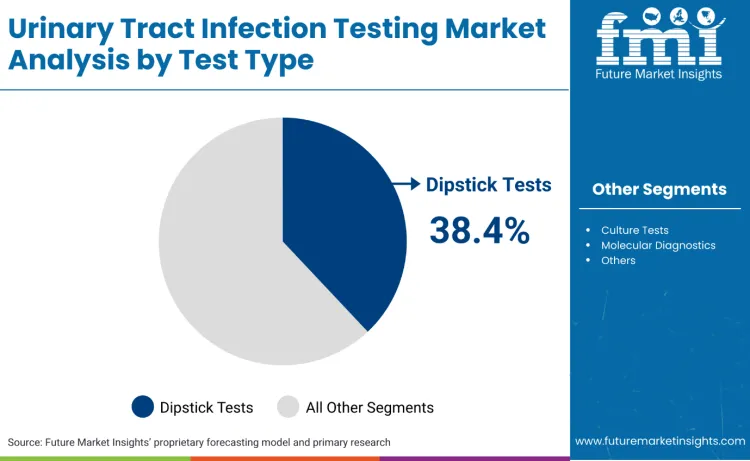
Dipstick tests account for 38.4%, driven by extensive use in primary care settings, emergency departments, and point-of-care applications. Culture tests hold 28.7%, supporting definitive diagnosis, antibiotic susceptibility testing, and complex infection analysis. Molecular diagnostics represent 19.3%, favored for rapid identification, pathogen detection, and antimicrobial resistance testing. Other testing methods contribute 13.6%, used in specialized applications and research settings.
Key Points
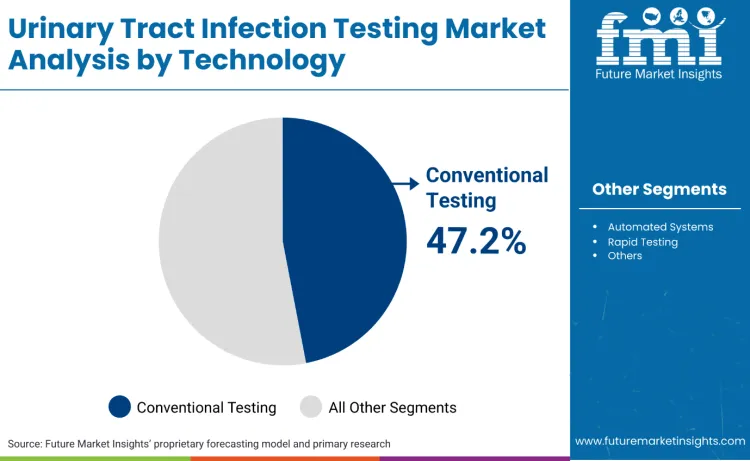
Conventional testing represents 47.2%, reflecting priority on applications requiring established protocols and cost-effective analysis. Automated systems account for 31.5%, suitable for high-throughput laboratories requiring standardized processing and reduced manual intervention. Rapid testing holds 21.3%, used for point-of-care applications requiring immediate results and clinical decision support.
Key Points
Demand in the urinary tract infection testing market arises from healthcare providers seeking to improve diagnostic accuracy, reduce testing costs, and meet clinical guidelines in applications where traditional methods prove insufficient. Activity is notable in hospital laboratories, outpatient clinics, and point-of-care facilities where diagnostic tests address infection detection, pathogen identification, and treatment guidance standards. Test selection centers on analytical performance, turnaround time, and cost efficiency features that support clinical decision-making without compromising diagnostic reliability.
Market uptake grows where evidence-based testing protocols are critical to patient care quality. In hospital and clinical settings, diagnostic tests must maintain accuracy specifications and turnaround time requirements to achieve treatment optimization and antibiotic stewardship compliance. Large-scale healthcare operations use dipstick and culture testing platforms to ensure diagnostic reliability during clinical decision processes, preventing treatment delays while maintaining cost-effective testing characteristics.
Healthcare providers depend on consistent testing performance to deliver infection detection, pathogen identification, and antimicrobial susceptibility capabilities comparable to established diagnostic protocols. Operators evaluate performance under diverse patient populations and clinical scenarios to ensure diagnostic accuracy through various healthcare delivery environments. Selection reflects how well a testing platform integrates with existing laboratory equipment, information systems, and clinical workflows common to modern healthcare operations.
Broader adoption is shaped by the complexity of healthcare reimbursement policies and specialized laboratory infrastructure requirements. Some applications experience cost constraints due to diagnostic equipment pricing fluctuations or reagent availability limitations, requiring careful technology selection and supplier management strategies. Regional differences in laboratory capacity and testing infrastructure affect where healthcare providers can access cost-effective diagnostics without triggering complex operational challenges.
Price considerations arise when accuracy requirements and turnaround time capabilities are needed to balance testing performance, operational costs, and total ownership expenses in facilities requiring consistent diagnostic specifications. Technology evaluation processes that assess analytical capabilities and operational efficiency extend testing selection timelines, influencing decisions about which platforms to deploy across diverse healthcare regions where cost requirements drive diagnostic approach decisions.
Global demand for urinary tract infection testing is increasing as healthcare systems expand diagnostic capacity while addressing patient care requirements, clinical accuracy, and treatment optimization application needs. Growth reflects rising use of automated testing systems, rapid diagnostic technologies, and point-of-care testing specifications across hospital laboratories, outpatient facilities, and primary care settings. Testing selection focuses on analytical performance, operational efficiency, and cost performance under various clinical scenarios. India records 5.8% CAGR, USA records 4.6% CAGR, Germany records 4.3% CAGR, Japan records 4.0% CAGR, and UK records 3.7% CAGR. Adoption remains driven by diagnostic requirements and clinical efficiency rather than volume expansion alone.
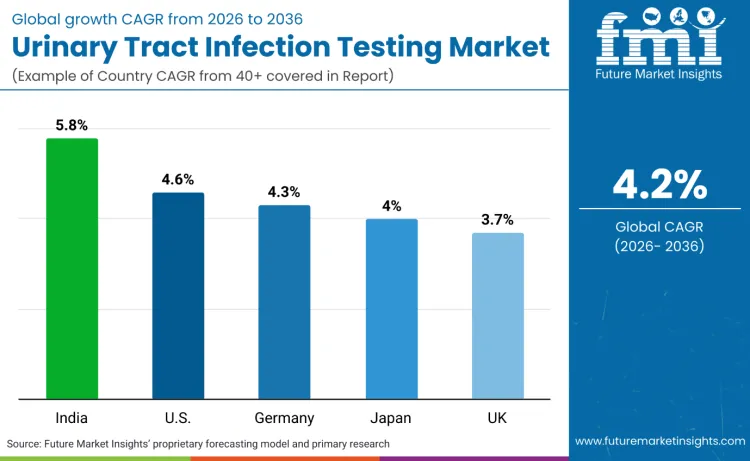
| Country | CAGR (2026-2036) |
|---|---|
| India | 5.8% |
| USA | 4.6% |
| Germany | 4.3% |
| Japan | 4.0% |
| UK | 3.7% |
Source: FMI’s proprietary forecasting model and primary research
The Indian market is being driven by the massive expansion of the Ayushman Bharat Digital Mission, which has integrated over 150,000 wellness centers with standardized diagnostic protocols. A key development in early 2026 is the CDSCO's fast-track approval for indigenous PCR-based UTI kits from domestic players like Mankind Pharma, aimed at providing affordable, high-sensitivity testing in rural settings. These localized kits are crucial for addressing the high prevalence of UTIs linked to diabetes and sanitation challenges. Driven by this aggressive infrastructure build-out, the Indian UTI testing market is set to grow at a 5.8% CAGR during the study period, leading the Asia-Pacific region in growth.
The 2026 global Urinary Tract Infection (UTI) testing market is undergoing a transition from traditional lab-based cultures, which often take 48 hours, to rapid molecular and AI-driven point-of-care (POC) diagnostics. This shift is driven by the urgent need for "diagnostic-driven antibiotic stewardship" to combat the escalating global threat of antimicrobial resistance (AMR).
The Indian market is being driven by the massive expansion of the Ayushman Bharat Digital Mission, which has integrated over 150,000 wellness centers with standardized diagnostic protocols. A key development in early 2026 is the CDSCO's fast-track approval for indigenous PCR-based UTI kits from domestic players like Mankind Pharma, aimed at providing affordable, high-sensitivity testing in rural settings. These localized kits are crucial for addressing the high prevalence of UTIs linked to diabetes and sanitation challenges. Driven by this aggressive infrastructure build-out, the Indian UTI testing market is set to grow at a 5.8% CAGR during the study period, leading the Asia-Pacific region in growth.
The Restraint: Despite infrastructure gains, high AMR rates for common antibiotics (like fluoroquinolones) in India mean that simple dipsticks are often insufficient. The market is restrained by the lack of affordable, rapid susceptibility testing that can be deployed at the primary care level.
In the USA, the primary driver is the 2025-2026 expansion of Medicare and private payer reimbursement for CLIA-waived molecular analyzers. This allows urgent care centers and even home-care providers to utilize sophisticated tools like the Roche Cobas Liat System for 20-minute pathogen identification. Furthermore, the FDA's tighter 2025 regulations on molecular assays have favored established leaders who can provide extensive resistance profiling alongside identification. Reflecting a mature market with high technological uptake, the USA UTI testing market is projected to grow at a 4.6% CAGR during the study period.
Germany’s market is characterized by the integration of Artificial Intelligence (AI) into automated urine sediment analyzers. With a shrinking specialized lab workforce, major German hospitals are adopting Siemens Healthineers’ AI-driven digital microscopy systems to automate the identification of bacteria and white blood cells. A critical 2026 driver is the "Digital Healthcare Act" (DVG) update, which incentivizes hospitals to use diagnostic data to prevent catheter-associated UTIs (CAUTIs). Within this efficiency-focused landscape, the German UTI testing market is expected to grow at a 4.3% CAGR during the study period.
In Japan, the world's most aged population is driving demand for tests that prevent the progression of simple UTIs into life-threatening sepsis or pyelonephritis. Moreover, the advancements have taken place and digital transformation in healthcare has come to the forefront. A major development is the 2025-2026 rollout of Sysmex Corporation’s flow cytometric analyzers specifically tailored for geriatric care facilities. These devices provide rapid, high-volume screening to identify asymptomatic bacteriuria in elderly patients, a major cause of hospital admissions in Japan. Supported by this demographic necessity, the Japanese UTI testing market is set to grow at a 4.0% CAGR during the study period.
The UK market is undergoing a major decentralization via the "Pharmacy First" initiative, which by early 2026 has successfully delivered over 600,000 UTI consultations in high-street pharmacies. A pivotal driver is the January 2026 NHS mandate for pharmacists to use "smartphone-read" dipsticks that provide digital evidence of infection before dispensing antibiotics. This policy aims to reduce the burden on General Practitioners (GPs) while maintaining strict antimicrobial stewardship. Supported by these public health shifts, the UK UTI testing market is set to grow at a 3.7% CAGR during the study period.
Quality assessment includes sensitivity performance, specificity validation, turnaround time compatibility, and integration with existing laboratory protocols. Procurement behavior reflects extensive performance validation, supply reliability requirements, and reliance on suppliers offering technical support and consistent testing specifications. Trends in the urinary tract infection testing market reflect emphasis on analytical quality and operational efficiency in hospital laboratories, outpatient clinics, and point-of-care applications.
Abbott Laboratories accounts for a leading position through integrated diagnostic testing operations designed to deliver analytical accuracy, operational reliability, and supply consistency capabilities. Roche Diagnostics competes by combining automated testing platforms with comprehensive quality assurance systems supporting clinical decision-making across diverse healthcare applications. Siemens Healthineers AG supports demand through specialized laboratory automation platforms aligned with efficiency and accuracy requirements. bioMérieux SA maintains relevance by supplying precision testing systems enhancing diagnostic accuracy and operational performance potential. Becton Dickinson and Company participates with diagnostic solutions emphasizing cost efficiency and clinical integration. Competitive differentiation centers on testing accuracy, supply reliability, technical specifications, and depth of clinical collaboration.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Test Type | Dipstick Tests; Culture Tests; Molecular Diagnostics; Others |
| Technology | Conventional Testing; Automated Systems; Rapid Testing; Others |
| End-User | Hospitals & Clinics; Diagnostic Laboratories; Home Care Settings; Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, USA, Germany, Japan, UK, and 40+ countries |
| Key Companies Profiled | Abbott Laboratories; Roche Diagnostics; Siemens Healthineers AG; bioMérieux SA; Becton Dickinson and Company; Others |
| Additional Attributes | Dollar sales by test type, technology, and end-user; performance in analytical accuracy and cost efficiency across hospital laboratories, point-of-care testing, and diagnostic facility matrices; testing quality improvement, operational efficiency enhancement, and clinical benefit under healthcare operations; impact on diagnostic accuracy, supply reliability, and cost management during clinical processes; compatibility with laboratory systems and quality control targets; procurement dynamics driven by healthcare facility expansion, quality improvement programs, and long-term diagnostic supply partnerships. |
The global urinary tract infection testing market is estimated to be valued at USD 52.3 billion in 2026.
The market size for the urinary tract infection testing market is projected to reach USD 78.9 billion by 2036.
The urinary tract infection testing market is expected to grow at a 4.2% CAGR between 2026 and 2036.
The key test types in the urinary tract infection testing market include dipstick tests, culture tests, molecular diagnostics, and other specialized diagnostic testing methods.
In terms of test type, the dipstick tests segment is set to command a 38.4% share in the urinary tract infection testing market in 2026.
Our Research Products

The "Full Research Suite" delivers actionable market intel, deep dives on markets or technologies, so clients act faster, cut risk, and unlock growth.

The Leaderboard benchmarks and ranks top vendors, classifying them as Established Leaders, Leading Challengers, or Disruptors & Challengers.

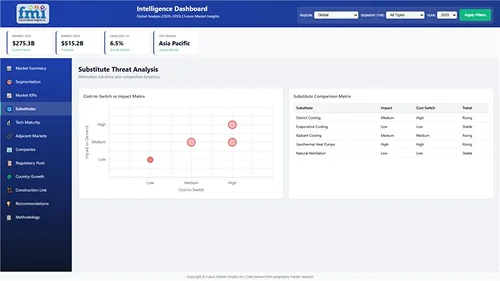
Locates where complements amplify value and substitutes erode it, forecasting net impact by horizon

We deliver granular, decision-grade intel: market sizing, 5-year forecasts, pricing, adoption, usage, revenue, and operational KPIs—plus competitor tracking, regulation, and value chains—across 60 countries broadly.

Spot the shifts before they hit your P&L. We track inflection points, adoption curves, pricing moves, and ecosystem plays to show where demand is heading, why it is changing, and what to do next across high-growth markets and disruptive tech

Real-time reads of user behavior. We track shifting priorities, perceptions of today’s and next-gen services, and provider experience, then pace how fast tech moves from trial to adoption, blending buyer, consumer, and channel inputs with social signals (#WhySwitch, #UX).
Partner with our analyst team to build a custom report designed around your business priorities. From analysing market trends to assessing competitors or crafting bespoke datasets, we tailor insights to your needs.
Supplier Intelligence
Discovery & Profiling
Capacity & Footprint
Performance & Risk
Compliance & Governance
Commercial Readiness
Who Supplies Whom
Scorecards & Shortlists
Playbooks & Docs
Category Intelligence
Definition & Scope
Demand & Use Cases
Cost Drivers
Market Structure
Supply Chain Map
Trade & Policy
Operating Norms
Deliverables
Buyer Intelligence
Account Basics
Spend & Scope
Procurement Model
Vendor Requirements
Terms & Policies
Entry Strategy
Pain Points & Triggers
Outputs
Pricing Analysis
Benchmarks
Trends
Should-Cost
Indexation
Landed Cost
Commercial Terms
Deliverables
Brand Analysis
Positioning & Value Prop
Share & Presence
Customer Evidence
Go-to-Market
Digital & Reputation
Compliance & Trust
KPIs & Gaps
Outputs
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE
Urinary Tract Infection (UTI) Treatment Market (UTI) Analysis - Size, Share, and Forecast 2025 to 2035
Self-urinary Infection Testing Market Size and Share Forecast Outlook 2025 to 2035
Complicated Urinary Tract Infections Treatment Market - Trends & Outlook 2025 to 2035
Uncomplicated Urinary Tract Infection Treatment Market Analysis by Penicillin, Quinolones, Sulfonamide, Nitrofuran, and Others Through 2035
Market Share Breakdown of Uncomplicated Urinary Tract Infection Treatment
Catheter Associated Urinary Tract Infections (UTI) Treatment Market - Demand & Forecast 2025 to 2035
Demand for Uncomplicated Urinary Tract Infection Treatment in Japan Size and Share Forecast Outlook 2025 to 2035
Demand for Uncomplicated Urinary Tract Infection Treatment in USA Size and Share Forecast Outlook 2025 to 2035
Pertussis Infection Testing Market
The Mosquito-borne Infections Testing Market is segmented by Test Type (Molecular Diagnostics, Immunoassay Systems, ELISA, Rapid Tests, Blot Systems and Evaluation Software, and Microspot Array Diagnostics), Indication (Malaria, Zika Virus, Chikungunya, Dengue, and Others), End User (Hospitals and Clinics, Private Commercial Laboratories, Academic and Research Centers, and Others), and Region. Forecast for 2026 to 2036.
Upper Respiratory Tract Infection Treatment Market
Hemotransmissive Infections Testing Market Size and Share Forecast Outlook 2025 to 2035
Nipah Virus (NiV) Infection Testing Market Insights by Test Type, End User, and Region through 2035
Arthropod-borne Viral Infections Testing Market Size and Share Forecast Outlook 2025 to 2035
Traction Battery Market Forecast and Outlook 2026 to 2036
Infection Control Market Size and Share Forecast Outlook 2025 to 2035
Urinary Collection Device Market Size and Share Forecast Outlook 2025 to 2035
Urinary Bag Market - Trends & Forecast 2025 to 2035
Infection Prevention Market is segmented by Product type and End User from 2025 to 2035
Urinary Antibacterial & Antiseptic Pharmaceuticals Market Analysis – Forecast 2025 to 2035

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.