About The Report
Rare Neurological Disease Treatment Market Forecast and Outlook 2026 to 2036
The rare neurological disease treatment market is expected to rise from USD 21.5 billion in 2026 to USD 43.7 billion by 2036, at a 7.4% CAGR. Expansion over this period is tied to the ability of late-stage neurological assets to progress from regulatory approval into stable, multi-year treatment use across small and clinically complex patient populations.
Commercial performance in rare neurology increasingly depends on execution factors, including supply continuity, treatment initiation logistics, and reimbursement durability over extended disease courses. Developers with prior experience in chronic neurological conditions show stronger uptake where launch planning incorporates long-term patient retention and monitoring requirements. Companies such as Biogen and Roche have structured rare neurology programs around lifecycle management and real-world treatment continuity rather than single-indication expansion.
From 2025 to 2026, growth coincided with portfolio rationalization across multiple rare neurology developers. Pipeline breadth narrowed as lower-probability programs were discontinued and capital was redirected toward assets approaching commercialization. Investment decisions favored later-stage neurological therapies with defined regulatory pathways, established specialist prescriber bases, and clearer access planning.
Rare neurological disease pipelines now exhibit higher concentration around fewer commercial assets. Launch readiness, manufacturing reliability, and access execution carry greater weight in portfolio prioritization, particularly in ultra-rare indications where supply disruption affects prescriber confidence and patient adherence. Firms such as Novartis and UCB maintain neurological portfolios centered on scalable production and specialist-led distribution models.
Treatment delivery characteristics influence demand persistence. Regimens that reduce procedural burden demonstrate higher continuation rates in progressive neurological conditions where mobility declines over time. Oral and simplified outpatient therapies extend treatment reach, while neurologist-led initiation and monitoring remain central to early prescribing decisions. Hospital and specialist channels continue to anchor initiation even as long-term treatment management increasingly occurs outside acute care settings.
“In rare neurological diseases, success is increasingly determined after approval. Therapies that combine clinical benefit with delivery models patients can sustain over years are the ones that ultimately define market leadership.”
- David Drennan, Senior Director, Drug Intelligence, Clarivate
Quick Stats for Rare Neurological Disease Treatment Market
- Rare Neurological Disease Treatment Market (2026): USD 21.5 billion
- Rare Neurological Disease Treatment Market Value (2036): USD 43.7 billion
- Rare Neurological Disease Treatment Market Forecast CAGR (2026 to 2036): 7.4%
- Leading Drug Class: Anti-depressants (26.0%)
- Leading Route of Administration: Oral (72.0%)
- Leading Distribution Channel: Hospital Pharmacies (43.0%)
- Leading Indication: Amyotrophic Lateral Sclerosis (ALS) (30.0%)
- Key Growth Countries: USA, Germany, Japan, South Korea, UK, China
- Key Players: Pfizer, Novartis, Merck, Johnson & Johnson, Bayer, GSK, Sanofi, AbbVie, Teva, Roche, and others
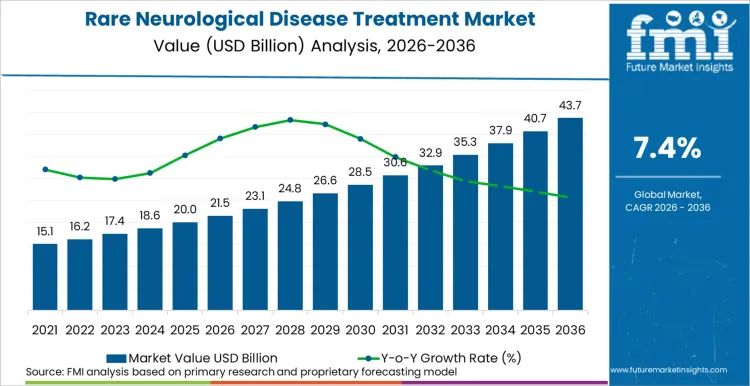
Rare Neurological Disease Treatment Market Key Takeaways
| Metric | Details |
|---|---|
| Market Value (2026) | USD 21.5 billion |
| Market Value (2036) | USD 43.7 billion |
| CAGR (2026 to 2036) | 7.4% |
Growth in rare neurological disease treatments reflects increased late-stage development activity across rare and ultra-rare indications, while commercial uptake depends on coordinated launch timing and payer-relevant clinical documentation that can support reimbursement decisions despite limited patient populations and trial sizes.
Neurology pipelines follow commercialization practices already established in chronic specialty care, with adoption drivers drawn from orphan drug strategies and long-term treatment management approaches used in complex chronic neurological indications.
Why is the Rare Neurological Disease Treatment Market Growing?
Demand growth reflects changes in diagnosis practices and treatment expectations occurring at the same time. Patients, caregivers, and clinicians increasingly expect earlier identification, simplified administration, and fewer facility-dependent steps, particularly in conditions where disease progression limits mobility and tolerance for repeated procedures. Demand favors formats and regimens that can be initiated by specialists and maintained with limited ongoing intervention, reinforcing uptake in institutional settings while supporting longer-term continuity outside acute care.
A further source of growth relates to changes in launch sequencing. Reliance-based approvals and cross-regulatory coordination are reducing time gaps between initial and follow-on approvals for certain therapies, allowing companies to generate earlier ex-US revenue and limit revenue loss associated with delayed market entry. Similar planning behavior is now evident in how sponsors structure evidence generation and market entry across neurological indications advancing through late-stage clinical trials.
On the supply side, manufacturers are reducing exposure to geopolitical and logistics risk by expanding domestically integrated manufacturing operations with greater process control and faster changeover capability. Novartis’ decision to invest USD 23 billion to expand U.S.-based manufacturing and R&D capacity illustrates how domestic capacity is increasingly treated as a factor supporting commercial execution rather than solely a procurement consideration.
Growth is also supported by lifecycle management decisions that adjust administration formats to expand treatable populations and reduce treatment complexity. Recent shifts toward subcutaneous and higher-concentration formulations have increased feasibility for home-based care while protecting long-term asset value. Similar approaches are visible across CNS treatment areas, where formulation changes, treatment and monitoring decisions, and neurological biomarker data increasingly influence prescribing behavior.
How the Rare Neurological Disease Treatment Market Segmented?
The market is segmented by drug class, route of administration, indication, and distribution channel. By drug class, offerings include anti depressants, anti psychotics, cholinesterase inhibitors, antagonists, and immunomodulators, reflecting varied therapeutic mechanisms. By route of administration, products are delivered through oral and injectable formats, addressing both chronic management and acute intervention needs. By indication, treatments target amyotrophic lateral sclerosis, spinal muscular atrophy, narcolepsy, chronic inflammatory demyelinating polyneuropathy, and Huntington disease, highlighting a focus on complex neurological disorders. By distribution channel, access is provided through hospital pharmacies, retail pharmacies, mail order pharmacies, and drug stores, covering institutional and outpatient care settings.
Why does ALS command the largest indication share in 2026?
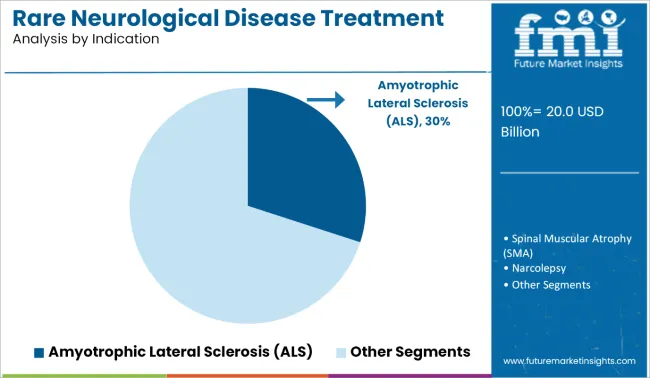
ALS leads with a 30.0% share in 2026. ALS is commercially attractive as treatment pathways increasingly allow multiple therapies to be used in parallel rather than as substitutes. In clinical practice, neurologists often layer approved disease-modifying agents with symptom-management drugs, extending treatment duration and increasing per-patient therapy value over time rather than triggering product replacement. This pattern is already visible in the ALS domain.
Biogen’s riluzole franchise continues to be used alongside newer agents rather than displaced, while Amylyx Pharmaceuticals positioned AMX0035 as an add-on therapy prior to its withdrawal, reflecting prescriber preference for combination use rather than monotherapy substitution. Similar expectations shaped development strategy at Ionis Pharmaceuticals, where antisense ALS programs are evaluated for compatibility with existing standards of care rather than as standalone replacements.
Why is Oral Drug Delivery the Preferred Route of Administration?
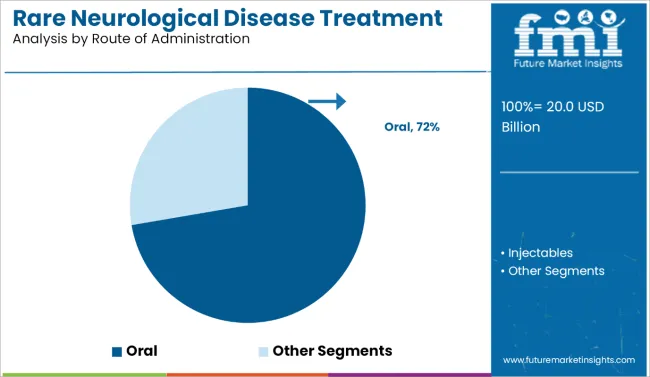
Oral delivery holds a 72.0% share in 2026 as rare-disease care is being de-medicalized, with manufacturers prioritizing adherence and convenience over facility-bound administration. The shift is reinforced by commercial execuistribution, reduce administration staffing requirements, and improve persistence where caregiver capacity is a constraint.
Evidence of this direction is reflected in the market impact of oral conversions that remove cold-chain infusion dependencies, making maintenance therapy compatible with home care and reducing discontinuation risk. These patterns increasingly mirror delivery redesign priorities visible in nucleic acid and gene therapies in neuromuscular disorders.
What renders Anti-Depressants the leading Drug Class?
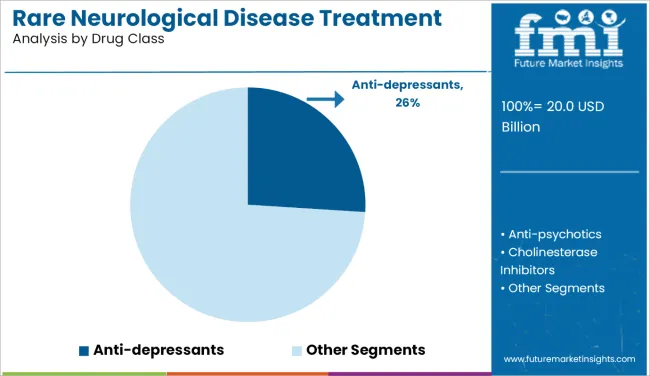
Anti-depressants lead with a 26.0% share in 2026 as a meaningful portion of demand is for symptomatic management linked to neurodegeneration rather than primary psychiatric use. In practice, SSRIs and SNRIs are increasingly used within ALS protocols to manage pseudobulbar affect and neuropathic supports adherence, especially in older patient cohorts.
Treatment portfolios and channel strategies frequently overlap with supportive care, including narcolepsy treatment and broader neurodegenerative disease treatment.
Section 5: What Trends are reshaping the Rare Neurological Disease Treatment Industry?
AI augmented regulatory review in the United Kingdom and South Korea is tightening review cycles and reducing uncertainty in rare neurological disease approvals. Automated data validation, protocol consistency checks, and parallel review workflows are shortening decision windows. This matters because orphan assets carry high development risk and limited patient pools, where launch delays materially erode returns. More predictable timelines improve ROI math and allow companies to sequence multi country filings with greater precision. Firms able to prepare manufacturing, clinical, and regulatory modules in parallel gain a structural advantage over peers using serial development models, improving speed to market and competitive positioning.
Why Is the Shift Toward End to End Domestic Manufacturing Accelerating
End to end domestic manufacturing footprints are expanding to reduce geopolitical exposure and protect supply continuity. Rare neurology therapies operate within narrow patient windows, where disruptions can trigger treatment discontinuation and loss of clinical momentum. Localized production improves predictability, shortens deviation response times, and reduces cross border logistics risk. This reliability strengthens confidence during reimbursement negotiations and hospital formulary reviews. Manufacturing location is therefore becoming part of the commercial value proposition, not just an operational choice. Companies with resilient domestic capacity are better positioned to support consistent patient access and defend long term market presence.
How Is Delivery Redesign Driving Outpatient Friendly Market Expansion
Delivery redesign toward outpatient friendly formats is emerging as a major commercial driver. Subcutaneous administration and high dose regimens reduce hospital dependence and lower treatment friction for patients and providers. This shift expands the addressable population by enabling treatment outside specialized centers. Improved convenience supports adherence and persistence, transforming demand from episodic hospital uptake into repeatable maintenance use. Lifecycle value is extended as therapies integrate more seamlessly into daily care pathways. Competitive advantage increasingly favors products that balance efficacy with ease of administration rather than relying solely on clinical differentiation.
Country-wise Analysis
Against the global CAGR of 7.4%, most countries are expanding at a faster pace, signaling structurally strong momentum in rare neurological disease treatments. China leads at 9.7%, supported by rapid clinical pipeline expansion and faster adoption of specialty therapies. Japan follows at 9.5%, driven by accelerated regulatory pathways and strong uptake of advanced neurology drugs. Germany posts 9.2%, reflecting reimbursement backed adoption and specialization in orphan care. The United Kingdom, at 8.6%, and South Korea, at 8.2%, both exceed the global rate due to regulatory modernization and innovation focused launches. The United States, at 7.6%, remains slightly above global growth, anchored in pipeline depth and early access programs.
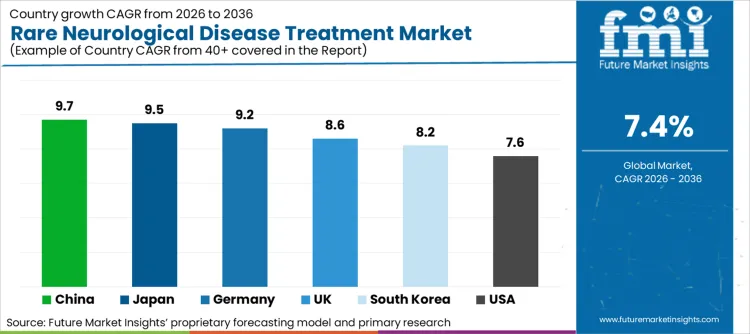
| Country | CAGR (2026-2036) |
|---|---|
| USA | 7.6% |
| Germany | 9.2% |
| Japan | 9.5% |
| South Korea | 8.2% |
| UK | 8.6% |
| China | 9.7% |
Why does USA remain the Market for Rare Neurological Disease Treatments?
USA continues to function as the primary reference market for rare neurological disease treatments, expanding at a 7.6% CAGR. Its role is defined less by volume and more by its ability to set regulatory, pricing, and evidence standards that shape downstream market behavior globally. A high concentration of orphan designations and late-stage neurology trials ensures that most first-in-class and first-to-market therapies establish their commercial benchmarks in the U.S. before international expansion.
From a demand perspective, U.S. growth is reinforced by strong diagnostic depth and specialist density, which enable earlier identification of rare neurological conditions compared to many other regions. However, scale is not automatic. Patient identification remains selective, and initiation often depends on structured neurologist-led protocols that concentrate early uptake within tertiary centers. This makes hospital-linked channels central to market access, even as maintenance therapy increasingly shifts toward outpatient and specialty pharmacy models.
What positions Germany as a High-growth yet Evidence-sensitive Market?
Germany is projected to grow at a 9.2% CAGR, reflecting strong reimbursement capacity alongside stringent benefit assessment expectations. The country’s rare neurological disease treatment demand is driven by well-developed specialist networks and early access to innovative therapies, but scaling is closely tied to how convincingly clinical benefit can be demonstrated relative to existing standards of care.
Unlike markets where access is negotiated centrally, Germany’s structure places early emphasis on comparative evidence and post-launch data generation. For rare neurological diseases, this creates both opportunity and constraint. Therapies that show clear functional or quality-of-life improvement can achieve meaningful uptake, while those with narrower or surrogate endpoints face slower expansion despite orphan status.
How is Japan accelerating Demand through Structural and Demographic Alignment?
Japan is likely to expand at a 9.5% CAGR, driven by a convergence of demographic pressure and structural realignment within pharmaceutical development and commercialization. An aging population increases the prevalence and recognition of neurological disorders, while healthcare delivery priorities increasingly favor simplified treatment pathways that reduce procedural burden on patients and caregivers.
From a market-structure standpoint, Japan is benefiting from tighter integration between global R&D programs and domestic launch strategies. This alignment reduces lag between international approvals and local availability, improving continuity of innovation flow. At the same time, portfolio refocusing by major manufacturers is redirecting capital toward fewer, higher-conviction neurological assets with clearer commercial pathways.
Why is South Korea emerging as a Speed-driven Rare Neurology Market?
South Korea is expected to grow at an 8.2% CAGR, with demand shaped by regulatory efficiency rather than sheer market size. The country’s healthcare and regulatory framework increasingly prioritizes faster review cycles and parallel evaluation processes, which reduce uncertainty for manufacturers planning early entry into the market.
This speed advantage allows South Korea to function as a commercially relevant early-access market for selected rare neurological therapies. Growth is not uniform across modalities. Treatments requiring complex logistics or highly specialized administration infrastructure face scaling limitations, particularly outside major urban centers.
How does UK balance access ambition with population constraints?
The United Kingdom is projected to grow at an 8.6% CAGR, shaped by a pragmatic approach to rare disease access within a constrained population base. While the UK has strong scientific and clinical expertise in neurology, ultra-rare indications often face limitations in domestic patient numbers, which restrict large-scale evidence generation.
To offset this, demand growth increasingly depends on real-world evidence integration and adaptive access mechanisms. Treatments that can demonstrate measurable benefit through post-launch monitoring and functional outcomes are better positioned to sustain reimbursement and expand utilization.
What is enabling China to post the fastest growth rate among key markets?
China is forecasted to expand at a 9.7% CAGR, making it the fastest-growing country in the rare neurological disease treatment landscape. This growth is underpinned by regulatory acceleration, earlier clinical trial activation, and a growing willingness to integrate innovative therapies into the healthcare system ahead of full reimbursement alignment.
Demand expansion is supported by improved diagnostic reach and increasing awareness of rare neurological conditions, particularly in urban centers. Faster regulatory pathways allow selected therapies to reach patients earlier, enabling real-world data accumulation that strengthens future access negotiations.
How Competitive is the Rare Neurological Disease Treatment Market?
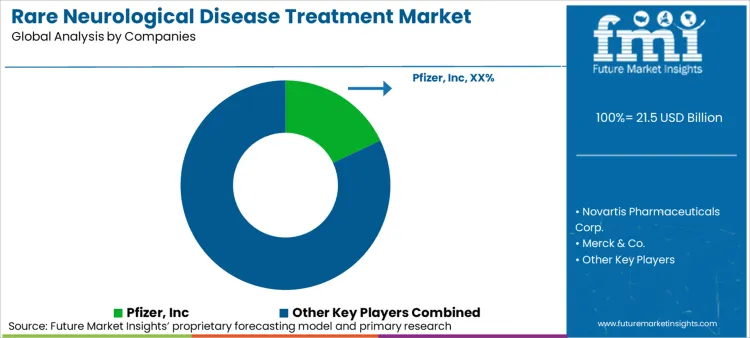
Competition in the rare neurological disease treatment market is shaped by execution capability rather than volume scale. With limited patient pools and specialist-led initiation, market leadership depends on how effectively companies translate regulatory approval into sustained, long-term therapy use. As a result, competitive advantage increasingly rests on delivery design, access navigation, and treatment continuity rather than pipeline breadth alone.
Large pharmaceutical companies such as Pfizer, Novartis, Merck & Co., and Johnson & Johnson maintain leadership by leveraging scale across regulatory execution, manufacturing reliability, and global access infrastructure. Their strength lies in absorbing high orphan development costs while sustaining post-launch investment in evidence generation and reimbursement engagement.
Mid-sized and therapy-focused players including AbbVie, Teva Pharmaceuticals, and F. Hoffmann-La Roche compete through depth within specific neurological pathways. These companies often focus on optimizing delivery formats, improving tolerability, and positioning therapies within combination regimens rather than pursuing first-in-class replacement strategies.
Distribution control has emerged as a key competitive lever. Companies with strong hospital-pharmacy engagement gain earlier initiation traction, while those with established specialty, retail, and mail-order pharmacy relationships are better positioned to protect adherence once patients transition to maintenance therapy. As oral administration dominates ongoing care, persistence and refill continuity increasingly determine lifetime value.
Key companies
- Pfizer Inc.
- Novartis Pharmaceuticals Corp.
- Merck & Co.
- Johnson & Johnson Services, Inc.
- Bayer AG
- GlaxoSmithKline, Inc.
- Sanofi S.A.
- AbbVie Inc. (Allergan, Inc.)
- Teva Pharmaceuticals
- F. Hoffmann-La Roche Ltd.
- Others
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 21.5 billion |
| Drug Class | Anti-depressants, Anti-psychotics, Cholinesterase Inhibitors, Antagonists, Immunomodulators |
| Route of Administration | Oral Formulations, Injectable Therapies, Subcutaneous Delivery, High-Concentration Formats |
| Indication Category | Amyotrophic Lateral Sclerosis (ALS), Spinal Muscular Atrophy (SMA), Narcolepsy, Chronic Inflammatory Demyelinating Polyneuropathy (CIDP), Huntington's Disease |
| Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Mail Order Pharmacies, Drug Stores, Specialty Pharmacy Networks |
| Treatment Approach | Disease-Modifying Therapies, Symptomatic Management, Combination Regimens, Add-On Therapies, Maintenance Treatment Protocols |
| Care Setting | Tertiary Centers, Outpatient Clinics, Home-Based Care, Specialist-Led Initiation, Neurologist-Coordinated Pathways |
| Commercial Focus | Launch Readiness, Supply Continuity, Treatment Initiation Logistics, Reimbursement Durability, Lifecycle Management, Real-World Evidence Generation |
| Manufacturing Strategy | Domestic Integrated Manufacturing, End-to-End Production Footprints, Localized Capacity Expansion, Process Control Systems, Rapid Changeover Capability |
| Regulatory Pathways | Orphan Drug Designations, AI-Augmented Review Processes, Accelerated Approval Mechanisms, Cross-Regulatory Coordination, Reliance-Based Approvals |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | United States, Germany, Japan, South Korea, United Kingdom, China, and 15+ countries |
| Key Companies Profiled | Pfizer Inc., Novartis Pharmaceuticals Corp., Merck & Co., Johnson & Johnson Services Inc., Bayer AG, GlaxoSmithKline Inc., Sanofi S.A., AbbVie Inc., Teva Pharmaceuticals, F. Hoffmann-La Roche Ltd. |
| Additional Attributes | Dollar sales by drug class and indication, regional demand trends, competitive landscape analysis, oral delivery dominance and adherence optimization, combination regimen economics, specialist prescriber engagement, hospital-to-outpatient transition models, treatment persistence and continuation rates, delivery redesign strategies, procedural burden reduction, diagnostic depth and identification timelines, reimbursement navigation frameworks, portfolio rationalization trends, late-stage asset prioritization, orphan drug execution strategies, evidence generation requirements, formulation lifecycle extensions, supply reliability impact on prescriber confidence, and patient retention mechanisms |
Rare Neurological Disease Treatment Market Segmentation
By Drug Class:
- Anti-depressants
- Anti-psychotics
- Cholinesterase Inhibitors
- Antagonists
- Immunomodulators
By Route of Administration:
- Oral
- Injectable
By Indication:
- Amyotrophic Lateral Sclerosis (ALS)
- Spinal Muscular Atrophy (SMA)
- Narcolepsy
- Chronic Inflammatory Demyelinating Polyneuropathy
- Huntington’s Disease
By Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Mail Order Pharmacies
- Drug Stores
By Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
Bibliography
- Biogen Inc. (2025, November 14). Biogen completes acquisition of Alcyone Therapeutics.
- Clarivate Plc. (2026, January 6). Clarivate identifies eleven potential blockbuster and transformative therapies in its 2026 Drugs to Watch report.
- National Medical Products Administration. (2025, October 14). Announcement on optimizing the review and approval of clinical trial applications for innovative drugs (30-day review pathway).
- Novartis. (2025, April 10). Novartis plans to expand its US-based manufacturing and R&D footprint with a total investment of $23B over the next 5 years.
- Sarepta Therapeutics, Inc. (2025, July 16). Sarepta Therapeutics announces strategic restructuring and pipeline reprioritization.
- USA Food and Drug Administration, Center for Drug Evaluation and Research. (2025). Advancing health through innovation: New drug therapy approvals 2024 (Annual report).
Frequently Asked Questions
How big is the global rare neurological disease treatment market?
The market is valued at USD 21.5 billion in 2026 and is projected to reach USD 43.7 billion by 2036.
What is the growth outlook over the next 10 years?
The rare neurological disease treatment market is forecasted to expand at a 7.4% CAGR from 2026 to 2036.
Which therapeutic areas or applications drive demand?
Demand is primarily driven by amyotrophic lateral sclerosis (ALS) with a 30.0% share
How does market behavior differ by region?
Growth is strongest in China (9.7%), Japan (9.5%), and Germany (9.2%), while the USA (7.6%) remains the commercialization anchor in this market.
What are the key risks, regulatory, or development constraints?
Key constraints include small patient populations that limit large datasets, rising payer scrutiny as therapy costs increase, and regulatory uncertainty tied to changing incentive structures for high-revenue orphan drugs.
Who are the leading companies in the Rare Neurological Disease Treatment Landscape?
Key companies include Pfizer, Novartis, Merck, Johnson & Johnson, Bayer, GSK, Sanofi, AbbVie, Teva, and Roche.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Class , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Class , 2026 to 2036
- Anti-depressants
- Anti-psychotics
- Cholinesterase Inhibitors
- Antagonists
- Immunomodulators
- Anti-depressants
- Y to o to Y Growth Trend Analysis By Drug Class , 2021 to 2025
- Absolute $ Opportunity Analysis By Drug Class , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Route of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Route of Administration, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Route of Administration, 2026 to 2036
- Oral
- Injectable
- Oral
- Y to o to Y Growth Trend Analysis By Route of Administration, 2021 to 2025
- Absolute $ Opportunity Analysis By Route of Administration, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Amyotrophic Lateral Sclerosis (ALS)
- Spinal Muscular Atrophy (SMA)
- Nacrolepsy
- Chronic Inflammatory Demyelinating Polyneuropathy
- Huntintong's Disease
- Amyotrophic Lateral Sclerosis (ALS)
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Hospital Pharmacies
- Retail Pharmacies
- Mail Order Pharmacies
- Drug Stores
- Hospital Pharmacies
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Drug Class
- By Route of Administration
- By Indication
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Pfizer, Inc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Novartis Pharmaceuticals Corp.
- Merck & Co.
- Johnson & Johnson Services, Inc.
- Bayer AG
- GlaxoSmithKline , Inc.
- Sanofi S.A.
- AbbVie Inc. (Allergan, Inc.)
- Teva Pharmaceuticals
- F. Hoffmann-La Roche Ltd.
- Others
- Pfizer, Inc
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Drug Class , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Drug Class
- Figure 6: Global Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Route of Administration
- Figure 9: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Indication
- Figure 12: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Distribution Channel
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Drug Class
- Figure 29: North America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Route of Administration
- Figure 32: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Indication
- Figure 35: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Distribution Channel
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Drug Class
- Figure 42: Latin America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Route of Administration
- Figure 45: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Indication
- Figure 48: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Drug Class
- Figure 55: Western Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Route of Administration
- Figure 58: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Indication
- Figure 61: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Drug Class
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Route of Administration
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Drug Class
- Figure 81: East Asia Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Route of Administration
- Figure 84: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Indication
- Figure 87: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Drug Class
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Route of Administration
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Drug Class , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Drug Class , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Drug Class
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Route of Administration
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Our Research Products

The "Full Research Suite" delivers actionable market intel, deep dives on markets or technologies, so clients act faster, cut risk, and unlock growth.

The Leaderboard benchmarks and ranks top vendors, classifying them as Established Leaders, Leading Challengers, or Disruptors & Challengers.

Locates where complements amplify value and substitutes erode it, forecasting net impact by horizon

We deliver granular, decision-grade intel: market sizing, 5-year forecasts, pricing, adoption, usage, revenue, and operational KPIs—plus competitor tracking, regulation, and value chains—across 60 countries broadly.

Spot the shifts before they hit your P&L. We track inflection points, adoption curves, pricing moves, and ecosystem plays to show where demand is heading, why it is changing, and what to do next across high-growth markets and disruptive tech

Real-time reads of user behavior. We track shifting priorities, perceptions of today’s and next-gen services, and provider experience, then pace how fast tech moves from trial to adoption, blending buyer, consumer, and channel inputs with social signals (#WhySwitch, #UX).
Partner with our analyst team to build a custom report designed around your business priorities. From analysing market trends to assessing competitors or crafting bespoke datasets, we tailor insights to your needs.
Supplier Intelligence
Discovery & Profiling
Capacity & Footprint
Performance & Risk
Compliance & Governance
Commercial Readiness
Who Supplies Whom
Scorecards & Shortlists
Playbooks & Docs
Category Intelligence
Definition & Scope
Demand & Use Cases
Cost Drivers
Market Structure
Supply Chain Map
Trade & Policy
Operating Norms
Deliverables
Buyer Intelligence
Account Basics
Spend & Scope
Procurement Model
Vendor Requirements
Terms & Policies
Entry Strategy
Pain Points & Triggers
Outputs
Pricing Analysis
Benchmarks
Trends
Should-Cost
Indexation
Landed Cost
Commercial Terms
Deliverables
Brand Analysis
Positioning & Value Prop
Share & Presence
Customer Evidence
Go-to-Market
Digital & Reputation
Compliance & Trust
KPIs & Gaps
Outputs
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

