About The Report
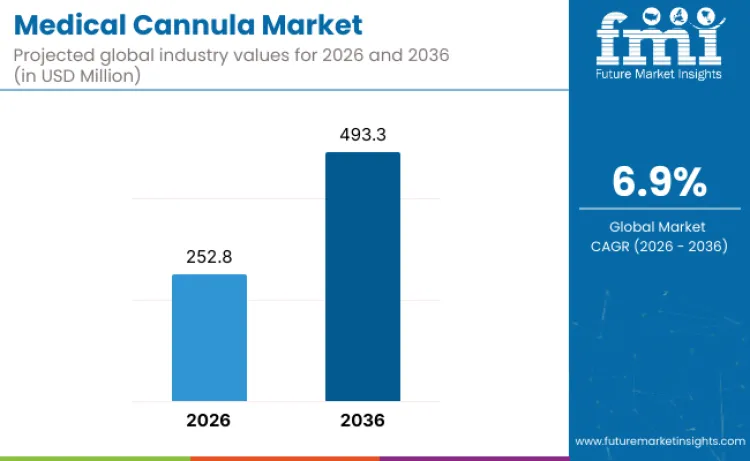
Medical cannula industry value is expected to reach USD 252.8 million in 2026, supported by rising use of minimally invasive procedures and continued expansion of healthcare capacity across multiple regions. As per FMI projections, revenue from medical cannula sales is set to grow at a 6.9% CAGR from 2026 to 2036, taking the market to USD 493.3 million by the end of the forecast period. Higher chronic disease burden and increasing outpatient procedure volumes are key factors supporting this growth outlook.
Hospitals and surgical centers are investing in improved cannula designs that enhance procedural control while meeting strict safety expectations. Manufacturers are also shifting from conventional metal formats toward biocompatible polymer based options, which can improve handling characteristics and support broader use across modern clinical workflows. These technology upgrades are encouraging suppliers to expand product portfolios with new materials, refined tip designs, and insertion features tailored to specific procedures.
Stronger focus on infection prevention and patient safety is influencing purchasing decisions, with clinicians selecting cannulas based on validated performance, gauge requirements, and coating options that support consistent outcomes. FMI estimates that ongoing evaluation of material performance and standardization needs is reinforcing demand for reliable, specification aligned devices. Growth opportunities remain strong in emerging markets, where infrastructure investment and wider access to surgical care are increasing procedure volumes, supporting steady uptake of cannulation products across diverse patient groups.
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 252.8 Million |
| Industry Value (2036) | USD 493.3 Million |
| CAGR (2026-2036) | 6.9% |
Source: Future Market Insights - analysis driven by proprietary forecasting models and primary research
Vascular access adoption is increasing as interventional cardiology and catheter based procedures expand, creating stronger demand for devices designed for reliable access and consistent performance. Improvements in insertion methods are strengthening procedural workflows by reducing discomfort and supporting higher first pass success, which appeals to clinicians focused on safety and efficiency. Wider use of polymer based cannulas in catheterization settings is also broadening applicability across specialties, since these materials can support smoother handling and more predictable performance in routine and complex interventions.
Clinical training and hospital education programs that link correct vascular access technique to better patient outcomes are reinforcing protocol based adoption and standardizing device selection in many care settings. In response, manufacturers are developing more specialized cannula designs for high complexity cardiovascular use cases, with product variants tailored to procedure specific demands. Advanced coating technologies are also being adopted more widely to support smoother insertion, maintain flow stability, and reduce friction related issues during access and device management, helping providers improve performance without sacrificing procedural effectiveness.
Global landscape is intricately segmented to address diverse requirements of the surgical device industry, categorizing sector by product type, medical procedure, gauge size, and end user applications. Structural division allows stakeholders to identify specific procedural targets, such as rising demand for flexible cannula designs in minimally invasive operations or preference for rigid configurations in emergency medical applications. By analyzing these segments, manufacturers can tailor production capabilities to meet distinct requirements of surgical specialists versus general hospital procurement. Segmentation also highlights shift from generic cannula approaches to procedure-specific interventional devices. As industry matures, granular segmentation becomes essential for understanding surgical pathways and regulatory compliance, ensuring product development aligns with evolving procedural guidelines.
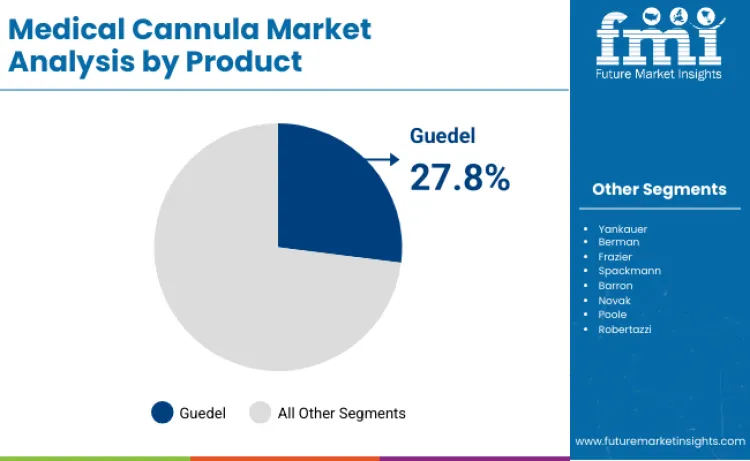
Guedel cannulas account for 27.8% of product share in 2026, driven by extensive clinical utilization in emergency airway management procedures. Leadership position is underpinned by regulatory approvals from medical authorities recognizing airway management therapeutic benefits. Segment benefits from continuous design improvements, such as anatomical curvature technology enhancing patient comfort during procedures. As per FMI's estimates, research focused on oropharyngeal airway optimization is renewing interest among emergency medicine manufacturers. Critical role in managing respiratory support applications further solidifies standing, as these devices ensure patent airway maintenance. As emergency medicine guidelines prioritize effective airway management, demand for medical-grade oropharyngeal cannulas is expected to remain robust.
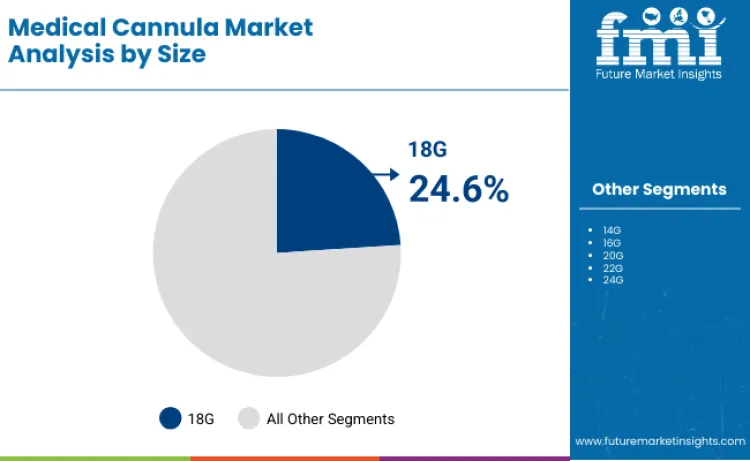
18G gauge size commands 24.6% share of sector in 2026, reflecting clinical preference for optimal flow rate versus insertion comfort balance. Dominance is linked to superior fluid administration capabilities and blood sampling efficiency compared to smaller gauge alternatives. FMI is of the opinion that hospitals rely on specialized 18G cannula technology to ensure rapid medication delivery and emergency fluid resuscitation. These configurations are essential for delivering concentrated medications and emergency treatments, driving priority status among emergency department protocols. Continuous pursuit of insertion ease ensures devices designed for rapid venous access remain primary choice. High clinical acceptance necessitates dynamic manufacturing capacity, bolstering sustained revenue streams for medical device suppliers.
Vascular access applications capture 74.2% of market share in 2026, owing to widespread intravenous therapy requirements across hospital settings. Dominance stems from clinical protocols mandating reliable venous access for medication administration, fluid management, and blood sampling procedures. Healthcare providers prefer these applications for their procedural versatility and patient safety benefits. Usage in treating emergency medical conditions drives continuous volume growth among acute care populations. As per FMI's projection, compatibility with various therapeutic interventions allows for combination procedures across different medical specialties. Manufacturers focusing on peripheral intravenous access formulations for these high-volume applications secure significant adoption among hospital procurement departments.
Demographic transition toward increased surgical procedure volumes is set to reshape sales landscapes by demanding specialized cannula configurations for diverse medical interventions. As surgical complexity trends evolve, hospitals access products with enhanced insertion characteristics for challenging anatomical access. Shift toward combination therapy procedures creates demand for multi-application cannula designs addressing multiple vascular access requirements simultaneously. Manufacturers failing to position cannulas as procedure-specific risk losing visibility among surgical procurement teams. Availability of specialized medical device options for chronic disease management drives complementary consumption patterns. Rise of robotic surgery applications stimulates innovation, with companies differentiating offerings through precision engineering integration.
Digital healthcare platform adoption is revolutionizing medical device procurement by enabling direct hospital ordering systems and inventory management solutions. Integration with electronic health records allows real-time tracking of cannula utilization patterns, triggering automated reorder protocols based on procedural volume data. Telemedicine consultations are driving standardized cannula selection protocols, with healthcare administrators specifying device requirements through digital procurement platforms. FMI is of the opinion that supply chain management systems are expanding rapidly, offering cost optimization and inventory control compared to traditional medical distribution channels. Mobile healthcare applications featuring device tracking and utilization monitoring create sustained engagement with hospital procurement programs.
Evolving regulatory frameworks are creating both opportunities and challenges for medical cannula manufacturers. Stricter biocompatibility standards demand higher manufacturing investments while improving clinical confidence in device safety profiles. Medical device approvals from regulatory bodies enable companies to market specific procedural benefits, differentiating products in competitive hospital markets. International harmonization of medical device regulations facilitates global market expansion for established manufacturers. As per FMI's estimates, clinical evidence requirements are increasing, forcing manufacturers to invest in human studies validating safety and efficacy claims.
Global landscape for medical cannula adoption is characterized by diverse healthcare systems, influenced by procedural standards and clinical practice protocols. Established markets prioritize evidence-based device selection and pharmaceutical-grade manufacturing standards, whereas emerging economies focus on cost-effective pricing and basic medical device accessibility. Asia-Pacific is emerging as rapid growth hub due to increasing surgical volumes and expanding hospital infrastructure development. Conversely, North American sectors pivot towards precision medicine and specialized procedural protocols. Government initiatives in developing nations supporting healthcare infrastructure development ensure sustained demand for medical devices across all demographic segments.
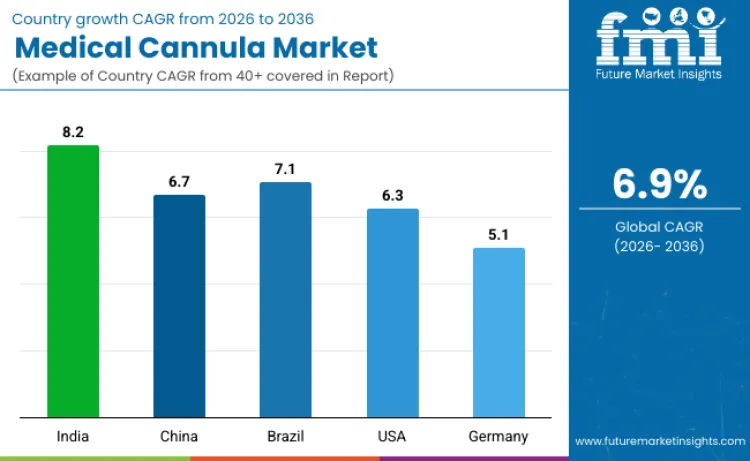
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 8.2% |
| China | 6.7% |
| Brazil | 7.1% |
| USA | 6.3% |
| Germany | 5.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Medical cannula sales in India are projected to rise at an 8.2% CAGR through 2036, supported by faster hospital expansion and growing procedure volumes across public and private care networks. Government reforms focused on hospital modernization and domestic medical device manufacturing are improving availability and procurement capacity, while expanding healthcare access is increasing adoption in both urban and rural facilities. FMI expects local production by global device manufacturers to reduce import reliance, improve supply stability, and support more competitive pricing. Continued development of emergency care services and upgrading of clinical equipment are also reinforcing demand, keeping India positioned as a high growth market for cannula use across multiple care settings.
China’s medical cannula demand is expected to grow at a 6.7% CAGR as healthcare modernization strengthens adoption of advanced surgical and interventional procedures. Investment in hospital infrastructure is improving distribution reach for specialized devices, while rising procedure volumes are pushing greater use of reliable vascular access products. Domestic manufacturers are expanding cost effective portfolios to serve a broader base of hospitals and clinics. FMI projects that policies promoting local medical device manufacturing will continue to accelerate uptake of indigenous brands and improve access across care centers, supporting steady market expansion.
Brazil’s medical cannula industry is forecast to expand at a 7.1% CAGR, mainly driven by higher surgical volumes in major urban areas and continued investment in healthcare delivery capacity. Public initiatives aimed at strengthening infrastructure and improving device access are encouraging hospitals to adopt more advanced procedural equipment. Partnerships between local distributors and international suppliers are improving product availability and supporting wider procurement. Economic improvement and broader insurance coverage are also increasing spending on hospital equipment. Strong regulatory oversight that emphasizes safety and performance is reinforcing confidence in advanced device use across clinical protocols.
Medical cannula sales in the United States are projected to increase at a 6.3% CAGR, supported by a mature healthcare system that continues adopting specialized devices to improve outcomes and efficiency. Growth is reinforced by rising procedural complexity and sustained expansion of minimally invasive techniques, which increase demand for precision access and management tools. Wider use of digital monitoring and standardized clinical pathways is supporting consistent utilization across care settings. FDA oversight and a strong evidence culture are encouraging ongoing product development and quality improvement. FMI expects patient safety and workflow efficiency priorities to remain central drivers, keeping the United States positioned as an innovation led market.
Germany is expected to see medical cannula revenue grow at a 5.1% CAGR, supported by strong emphasis on safety, validated performance, and modern surgical practices. Demand is strengthened by wider attention to patient protection standards and consistent protocol use in medical interventions. Adoption of precision surgery approaches is contributing to growth in specialized segments where high quality devices are required. FMI estimates that established medical device firms and rigorous clinical validation practices help maintain strong product standards. Continued focus on evidence-based medicine and regulatory compliance is expected to sustain Germany as a quality focused market for medical cannula solutions.
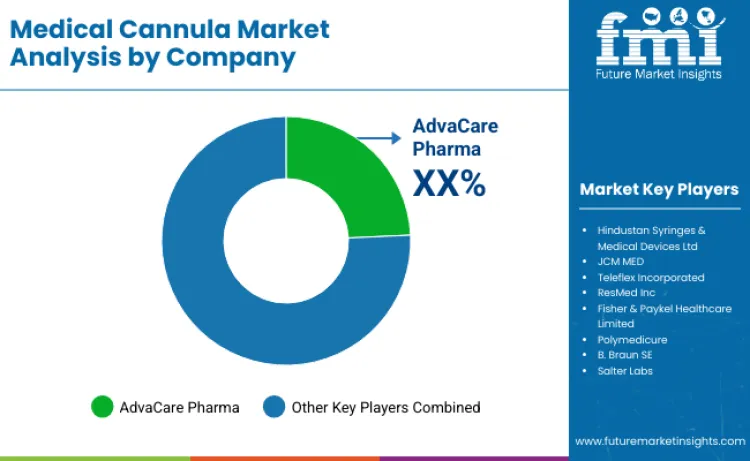
Competition in the medical cannula market is increasingly shaped by clinical proof, product reliability, and the ability to meet procedure specific requirements across diverse care settings. Many suppliers are emphasizing clinical validation to strengthen differentiation and support hospital procurement decisions. Companies such as AdvaCare Pharma and Teleflex Incorporated are investing in research collaborations with medical institutions to evaluate device performance, safety, and usability in real clinical environments.
A key product strategy is the development of specialized cannula ranges that help clinicians select the right configuration based on access type, patient profile, and procedural complexity, supporting more consistent outcomes and smoother workflow adoption. Vendors are also forming closer partnerships with healthcare providers to align devices with routine clinical protocols, training needs, and standardization goals. Innovation is focused on advanced materials and design improvements that enhance biocompatibility, handling, and insertion performance while maintaining safety expectations. Deal activity remains measured, with acquisitions often targeting component suppliers to secure quality, manage costs, and reduce supply risk in competitive tenders. FMI expects companies with strong distribution reach and sustained clinical research investment to retain an advantage, including AdvaCare Pharma, which benefits from global channel presence and continued focus on device development.
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Product Segments | Guedel; Yankauer; Berman; Frazier; Spackmann; Barron; Novak; Poole; Robertazzi; Bellucci; Brackman; Karman; Others |
| Procedure Categories | Vascular Access; Surgery; Intubation; Lipofilling; Suction; Irrigation; Biopsy; Insufflation; Hysterography; Insemination; Cardioplegia |
| Size Categories | 14G; 16G; 18G; 20G; 22G; 24G; 26G |
| Material Types | Latex; Latex-Free; Neoprene |
| End User Categories | Hospitals; Ambulatory Surgical Centers; Clinics; Other |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Key Countries | India, China, Brazil, USA, Germany |
| Key Companies Profiled | AdvaCare Pharma; Hindustan Syringes & Medical Devices Ltd; JCM MED; Teleflex Incorporated; ResMed Inc; Fisher & Paykel Healthcare Limited; Polymedicure; B. Braun SE; Salter Labs; ICU Medical, Inc. |
| Additional Attributes | Dollar sales measured for medical cannulas used in surgical procedures and medical interventions, specified by product type, gauge size, material composition, procedural application focus, end user preference, distribution model, and compliance alignment with evolving medical device standards and safety regulations. |
The global medical cannula market is valued at USD 252.8 million in 2026.
The market is projected to grow at a CAGR of 6.9% from 2026 to 2036, reaching USD 493.3 million by 2036.
Demand is primarily driven by vascular access procedures, which account for 74.2% share, across hospitals and ambulatory surgical centers.
Key barriers include pricing pressure, strict biocompatibility regulations, infection control requirements, and procurement sensitivity in cost-focused healthcare systems.
Regulatory approvals ensure product safety and standardization, while reimbursement frameworks indirectly influence procurement volumes through hospital cost controls.
Our Research Products

The "Full Research Suite" delivers actionable market intel, deep dives on markets or technologies, so clients act faster, cut risk, and unlock growth.

The Leaderboard benchmarks and ranks top vendors, classifying them as Established Leaders, Leading Challengers, or Disruptors & Challengers.

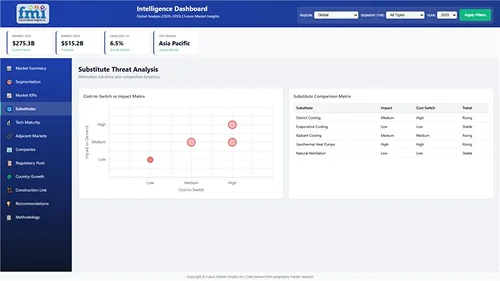
Locates where complements amplify value and substitutes erode it, forecasting net impact by horizon

We deliver granular, decision-grade intel: market sizing, 5-year forecasts, pricing, adoption, usage, revenue, and operational KPIs—plus competitor tracking, regulation, and value chains—across 60 countries broadly.

Spot the shifts before they hit your P&L. We track inflection points, adoption curves, pricing moves, and ecosystem plays to show where demand is heading, why it is changing, and what to do next across high-growth markets and disruptive tech

Real-time reads of user behavior. We track shifting priorities, perceptions of today’s and next-gen services, and provider experience, then pace how fast tech moves from trial to adoption, blending buyer, consumer, and channel inputs with social signals (#WhySwitch, #UX).
Partner with our analyst team to build a custom report designed around your business priorities. From analysing market trends to assessing competitors or crafting bespoke datasets, we tailor insights to your needs.
Supplier Intelligence
Discovery & Profiling
Capacity & Footprint
Performance & Risk
Compliance & Governance
Commercial Readiness
Who Supplies Whom
Scorecards & Shortlists
Playbooks & Docs
Category Intelligence
Definition & Scope
Demand & Use Cases
Cost Drivers
Market Structure
Supply Chain Map
Trade & Policy
Operating Norms
Deliverables
Buyer Intelligence
Account Basics
Spend & Scope
Procurement Model
Vendor Requirements
Terms & Policies
Entry Strategy
Pain Points & Triggers
Outputs
Pricing Analysis
Benchmarks
Trends
Should-Cost
Indexation
Landed Cost
Commercial Terms
Deliverables
Brand Analysis
Positioning & Value Prop
Share & Presence
Customer Evidence
Go-to-Market
Digital & Reputation
Compliance & Trust
KPIs & Gaps
Outputs
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE
The medical tourism industry in Thailand is segmented by Treatment Type (Cosmetic Surgery, Dental Treatment, Cardiac Surgery, Orthopedics, Infertility Treatment, Others), Service Provider (Private Hospitals, Public Hospitals, Clinics), and Region Forecast for 2026 to 2036.
The Medical Device Packaging Market is segmented by Material, Class, Application, Packaging Format (Bags & Pouches, Films & Wraps, Lids, Rollstock (Top and Bottom, Forming Webs), Containers, Trays, Stick Packs, Clamshell, Blister Packs, Boxes, Flow Wraps) and Region. Forecast for 2026 to 2036.
Medical-Grade PCR Packaging Market Size and Share Forecast Outlook 2026 to 2036
Medical Tourism Market Size and Share Forecast Outlook 2026 to 2036
Medical Cyclotron Market Trends and Growth Forecast 2026 to 2036
Medical Carts Market Forecast and Outlook 2026 to 2036
Medical Plastics Market Size and Share Forecast Outlook 2026 to 2036
Medical Devices Surface Active Coatings Market Size and Share Forecast Outlook 2026 to 2036
Medical Sharps Market Size and Share Forecast Outlook 2026 to 2036
Medical Specialty Bag Market Size and Share Forecast Outlook 2026 to 2036
Medical Adhesive-Related Skin Injury (MARSI) Treatment Market Analysis – Size, Share, and Forecast Outlook 2025 to 2035
Medical Silicone Radiopaque Vascular Ties Market Size and Share Forecast Outlook 2025 to 2035
Medical Indoor Air Purifier Market Size and Share Forecast Outlook 2025 to 2035
Medical Eye Shield Film Market Size and Share Forecast Outlook 2025 to 2035
Medical Far Infrared Therapy Device Market Size and Share Forecast Outlook 2025 to 2035
Medical Latex Protective Suit Market Size and Share Forecast Outlook 2025 to 2035
Medical Activated Carbon Dressing Market Size and Share Forecast Outlook 2025 to 2035
Medical Coated Roll Stock Market Size and Share Forecast Outlook 2025 to 2035
Medical Billing Outsourcing Market Size and Share Forecast Outlook 2025 to 2035
Medical Pressure Mapping System Market Size and Share Forecast Outlook 2025 to 2035

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.