Lung Cancer Therapeutics Market
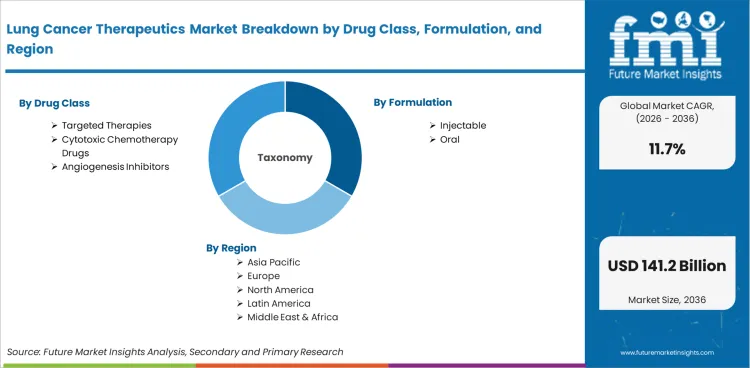
The Lung Cancer Therapeutics Market is segmented by Drug Class (Cytotoxic Chemotherapy Drugs, Targeted Therapies, Angiogenesis Inhibitors), Formulation (Injectable, Oral), Molecule Type (Small Molecule, Large Molecule), Cancer Type (Non Small Cell Lung Cancer, Small Cell Lung Cancer), Distribution Channel (Institutional Sales, Retail Sales), and Region. Forecast for 2026 to 2036.
Lung Cancer Therapeutics Market Size, Market Forecast and Outlook By FMI
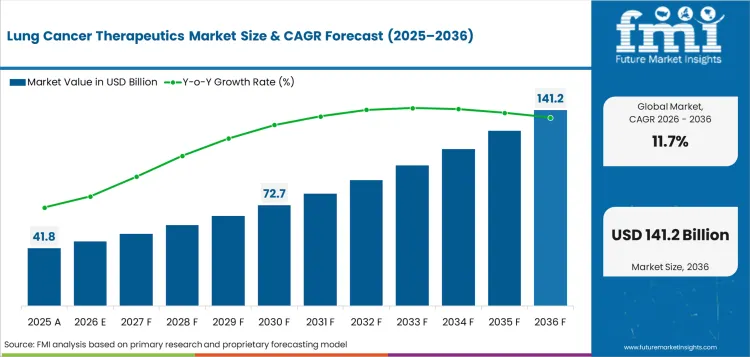
The lung cancer therapeutics market was valued at USD 41.8 billion in 2025. The market is forecast to reach USD 46.7 billion by the end of 2026. Demand is forecast to expand at a CAGR of 11.7% between 2026 and 2036 and reach USD 141.2 billion by 2036. Targeted Therapies segment is anticipated to lead with a 91.2% drug class share in 2026 and by formulation, injectable formulations is anticipated to be leading with a 61.0% share.
Summary of the Lung Cancer Therapeutics Market
- Demand and Growth Drivers
- Biomarker-based treatment selection supports demand for targeted therapies as oncologists match therapy choice with mutation status.
- Immunotherapy combinations raise institutional drug value as hospitals manage advanced NSCLC treatment through supervised protocols.
- Oral kinase inhibitor use expands repeat prescription demand as patients stay on maintenance treatment across longer care cycles.
- Product and Segment View
- Targeted Therapies are expected to account for 91.2% share in 2026 as mutation testing guides treatment choice across advanced NSCLC cases.
- Injectable formulations are projected to represent 61.0% share as infused biologics keep hospital-based dosing central to lung cancer care.
- Small Molecule demand is shaped by kinase inhibitor use across EGFR and ALK-positive patients requiring repeat therapy cycles.
- Geography and Competitive Outlook
- India is expected to record a CAGR of 17.6% through 2036, supported by broader oncology access and diagnosis depth.
- China is projected to expand at 17.0% CAGR by 2036, as large lung cancer incidence raises therapy demand.
- Companies combining immunotherapy depth with biomarker-directed portfolios strengthen their market position during the forecast period.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant at FMI says, “Lung cancer treatment now guides evidence-based therapy selection for oncology teams seeking fewer failed cycles. Treatment plans need biomarker records with reimbursement clarity. Clinical evidence and oncology access can reduce therapy switching risk under branded regimen cost pressure.”
- Lung Cancer Therapeutics Market Value Analysis
- The lung cancer therapeutics market is moving from broad chemotherapy use toward a structured precision oncology base.
- Mutation testing across EGFR, ALK, ROS1 and related NSCLC pathways strengthens treatment planning.
- Wider use of lung cancer diagnostics is supporting advanced therapy planning across oncology care.
- Therapy records meeting reimbursement and hospital protocol requirements favor adoption across high-cost oncology care.
Lung cancer therapy is shifting toward earlier, more targeted and more mechanism-specific treatment. FDA stated in August 2024 that lazertinib with amivantamab was approved for first-line EGFR-mutated advanced NSCLC. That gives oncologists another mutation-specific combination route beyond single-agent EGFR inhibition. FDA granted accelerated approval to tarlatamab-dlle in May 2024 for extensive-stage small cell lung cancer after platinum-based chemotherapy. Later-line small cell lung cancer therefore gains a new immunotherapy mechanism.
FDA approved durvalumab in August 2024 for use with chemotherapy before surgery and as adjuvant therapy after surgery in eligible resectable NSCLC. Perioperative treatment expands the commercial window into earlier disease stages. FDA also approved osimertinib with platinum-based chemotherapy in February 2024 for advanced EGFR-mutated NSCLC. Combination intensification is likely to shape first-line prescribing where physicians seek stronger disease control. Amgen’s DeLLphi-301 data adds commercial support for DLL3-targeted T-cell engagement.
Lung Cancer Therapeutics Market Definition
The lung cancer therapeutics market includes prescription drug therapies used to treat non small cell lung cancer and small cell lung cancer. Scope includes cytotoxic chemotherapy drugs, targeted therapies, angiogenesis inhibitors, monoclonal antibodies, kinase inhibitors, and approved combination regimens. Diagnostic-only products, radiation systems, surgery devices, and supportive care drugs without direct antitumor action are outside the scope.
Lung Cancer Therapeutics Market Inclusions
Market scope covers all commercially traded lung cancer therapeutic products categorized by drug class, including cytotoxic chemotherapy drugs, targeted therapies, and angiogenesis inhibitors; formulation, including injectable and oral therapies; molecule type, including small molecule and large molecule drugs; cancer type, including non small cell lung cancer and small cell lung cancer; and distribution channel, including institutional sales and retail sales. Revenue scope covers 2026 to 2036. Main participants include pharmaceutical companies, biotechnology firms, oncology drug manufacturers, hospital oncology departments, cancer treatment centers, specialty clinics, retail pharmacies, oncology distributors, clinical research organizations, payers, regulatory bodies, and healthcare providers.
Lung Cancer Therapeutics Market Exclusions
Scope does not include lung cancer screening tests, standalone molecular diagnostic panels, radiation oncology equipment, thoracic surgery devices, or palliative drugs without direct cancer treatment action.
Lung Cancer Therapeutics Market Research Methodology
- Primary Research: FMI analysts interviewed oncology specialists, hospital pharmacy teams, cancer treatment centers, and specialty drug channel participants in major markets.
- Desk Research: Desk review combined data from regulatory approvals, clinical treatment pathways, company oncology portfolios, and cancer burden references.
- Market sizing and forecasting: Market sizing added therapy use across treatment classes and country access levels from the bottom up.
- Data validation: Cross-checked every three months against therapy approvals, drug class mix, reimbursement patterns, and company portfolio coverage.
Why is the Lung Cancer Therapeutics Market Growing?
- Oncology teams are using molecular testing earlier to reduce ineffective therapy sequencing before advanced treatment begins.
- Treatment partners gain value as lung cancer regimens combine biologics and mutation-specific small molecules.
- Manufacturers are investing in drugs with tighter patient selection and better evidence packages for hospital approval committees.
Global lung cancer burden keeps therapy demand active across public and private oncology systems. Large patient volumes create steady treatment planning needs across first-line therapy and maintenance care. Relapse management requires planned sequencing as oncology teams manage recurring disease risk over time. Drug makers gain from therapies that fit biomarker pathways and reduce unplanned regimen changes. Hospitals need strong clinical evidence to justify reimbursement and protocol placement.
Regulatory approvals are expanding the number of mutation-defined treatment options available to oncologists. FDA approved osimertinib with platinum-based chemotherapy in February 2024 for EGFR-mutated locally advanced or metastatic NSCLC. The FDA approved taletrectinib in June 2025 for adults with locally advanced or metastatic ROS1-positive NSCLC. FDA approvals reinforce the role of EGFR-mutated lung cancer treatment in advanced NSCLC planning. Payers will expect stronger patient selection evidence as therapy costs rise.
Market Segmentation Analysis
- Targeted Therapies are estimated to capture 91.2% share of the Drug Class category in 2026 as biomarker-led prescribing keeps treatment selection focused on defined mutation profiles.
- Injectable formulations are projected to represent 61.0% of the Formulation segment in 2026 due to higher use across immunotherapy and biologic regimens.
- Small Molecule therapies are projected to hold 56.0% share by Molecule Type in 2026 with strong relevance across kinase inhibitor pathways.
- Non Small Cell Lung Cancer is expected to account for 87.4% of Cancer Type share in 2026 as therapy use is broad across major clinical subtypes.
- Institutional Sales are projected to secure 68.0% share of the Distribution Channel segment in 2026 because hospital cancer care is central to administration and monitoring.
Lung cancer therapeutics are divided into five primary segment groups based on drug class, formulation, molecule type, cancer type, and distribution channel. Drug class includes Cytotoxic Chemotherapy Drugs, Targeted Therapies, and Angiogenesis Inhibitors. Formulation includes Injectable and oral. Molecule type includes small molecule and large molecule. Cancer type includes non-small cell lung cancer and Small Cell Lung Cancer. Distribution channel includes Institutional Sales and Retail Sales.
Insights into the Targeted Therapies Drug Class Segment
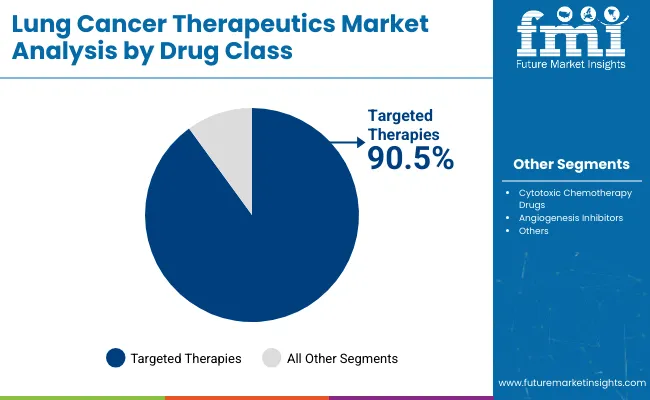
- In 2026, Targeted Therapies are expected to make up 91.2% of the Drug Class segment. Biomarker testing guides physicians toward mutation-specific drugs across advanced NSCLC treatment pathways.
- Chemotherapy and angiogenesis inhibitors address selected treatment needs. Their use is stronger in combination regimens and patients without actionable mutations.
Insights into the Injectable Formulation Segment
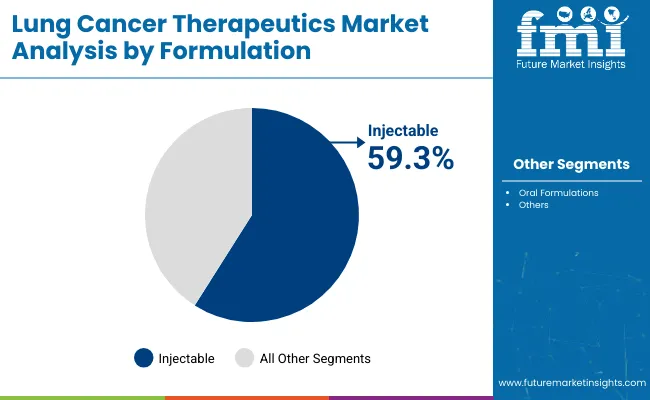
- Injectable therapies are projected to account for 61.0% of the Formulation segment in 2026. Infused immunotherapy and biologic regimens require hospital supervision across high-cost oncology care.
- Oral options address long-term treatment needs for selected patients. Their value improves in maintenance care and mutation-confirmed kinase inhibitor use.
Insights into the Small Molecule Molecule Type Segment
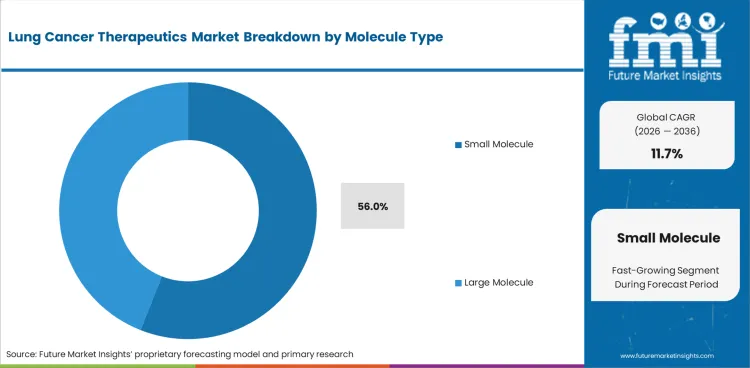
- Small Molecule drugs are estimated to account for 56.0% share in 2026. Oral kinase inhibitors carry strong use across EGFR and ALK-positive lung cancer cases.
- Large molecule therapies address immunotherapy-based treatment pathways. Their use is strongest in infused biologics and checkpoint inhibitor regimens across advanced disease.
Insights into the Non Small Cell Lung Cancer Cancer Type Segment
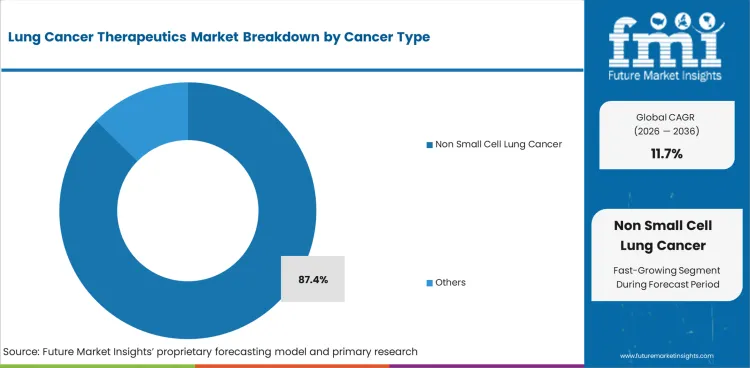
- Non Small Cell Lung Cancer is expected to represent 87.4% share in 2026. NSCLC has broader therapy options across targeted drugs and immune checkpoint regimens.
- Small Cell Lung Cancer treatment demand is more concentrated. Therapy value improves in extensive-stage disease and maintenance treatment settings.
Insights into the Institutional Sales Distribution Channel Segment
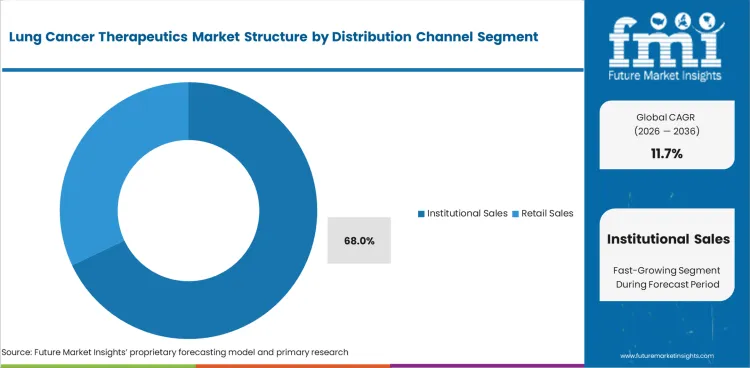
- Institutional Sales are projected to hold 68.0% share in 2026. Hospitals prefer direct therapy control for infused drugs and reimbursement documentation.
- Retail sales support oral therapy access for selected patients. Specialty pharmacies serve maintenance prescriptions and home delivery needs.
Lung Cancer Therapeutics Market Drivers, Restraints, and Opportunities
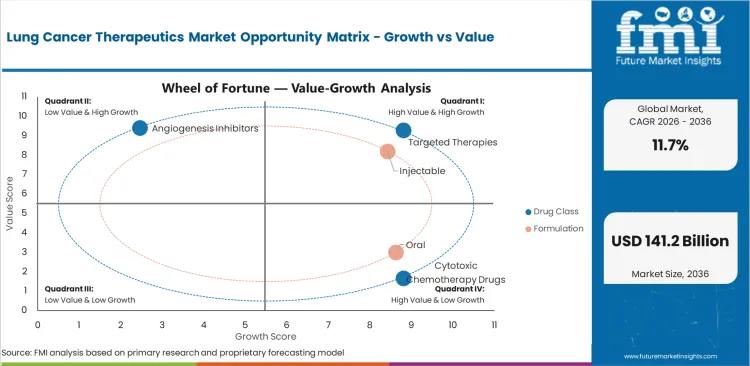
- Molecular profiling strengthens structured demand for targeted therapies across EGFR and ALK patient groups.
- ROS1-positive patients add demand for mutation-specific lung cancer treatment within advanced oncology care.
- High drug cost increases pricing pressure for payers managing branded immunotherapy and newer kinase inhibitor regimens.
- Subcutaneous biologic formats improve service opportunity by reducing infusion chair pressure in busy cancer centers.
Lung cancer therapeutics demand expands as therapy selection becomes more closely aligned with biomarker evidence. FDA approved atezolizumab and hyaluronidase-tqjs for subcutaneous injection in September 2024 across approved adult atezolizumab indications. The FDA approval gives hospitals another administration option for immunotherapy patients. Cancer centers can improve scheduling flexibility if subcutaneous formats reduce infusion time. Suppliers with injectable and alternate dosing formats can serve hospitals facing treatment capacity limits.
Tarlatamab Access Is Forcing Price Reassessment
Tarlatamab creates a clear price-pressure signal in the lung cancer therapeutics market because payers are testing whether late-line clinical value can justify premium biologic pricing. Canada’s Drug Agency reported that tarlatamab’s submitted price was CAD 1,545 per 1 mg vial and CAD 15,450 per 10 mg vial. Its reanalysis found an 85.6% price reduction would be needed to reach CAD 222 per 1 mg vial and CAD 2,220 per 10 mg vial at a CAD 50,000/QALY threshold. The reanalysis points to a wider gap between launch pricing and reimbursement acceptability for novel small-cell lung cancer therapies.
ALK Inhibitor Costs Keep Payer Budgets Elevated
Advanced ALK-positive NSCLC shows high recurring drug-cost exposure in lung cancer therapeutics. A September 2025 JMCP study of 696 patients reported total per-patient-per-month costs of USD 28,216 and average 30-day ALK TKI supply costs of USD 17,766. The cost figures keep pricing pressure concentrated at launch and through treatment sequencing. Only 24.3% of patients moved to another ALK TKI after first-line therapy. Manufacturers now need durability and tolerability to protect pricing as strongly as response data. Line-of-therapy positioning influences how well pricing can be defended over time.
Analysis of Lung Cancer Therapeutics Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| India | 17.6% |
| China | 17.0% |
| South Korea | 15.4% |
| Germany | 13.2% |
| Australia | 10.4% |
| United States | 6.6% |
| Japan | 8.4% |
Source: Future Market Insights, 2026.
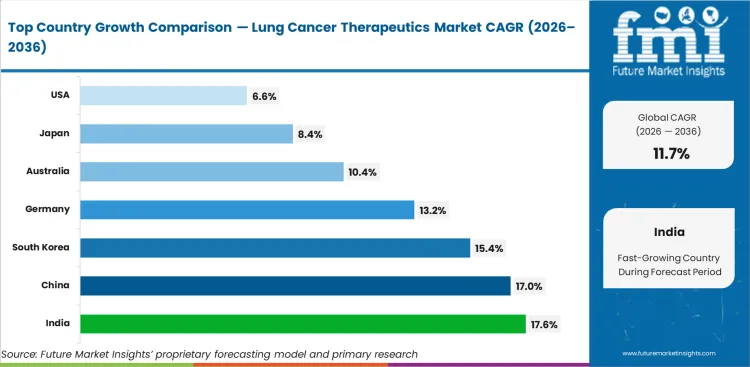
Lung Cancer Therapeutics Market CAGR Analysis By Country
- India is projected to record 17.6% CAGR over the forecast period, backed by broader oncology access and stronger diagnostic capacity.
- China is expected to expand at 17.0% CAGR between 2026 and 2036 as its large lung cancer caseload raises targeted therapy use.
- South Korea is forecast to grow at 15.4% CAGR during the assessment period, as organized cancer centers improve biomarker-based treatment access.
- Germany is expected to advance at 13.2% CAGR by 2036, helped by reimbursement systems supporting high-value oncology drugs.
- Australia is projected to rise at 10.4% CAGR across the ten-year outlook as screening rollout improves specialist treatment referral.
- United States is forecast at 6.6% CAGR over the same period, as market maturity limits percentage expansion despite a large, treated base.
- Japan is expected to post 8.4% CAGR from 2026 to 2036 as older patient burden sustains demand for advanced oncology therapies.
Demand for lung cancer therapeutics is forecast to rise at 11.7% CAGR from 2026 to 2036. Country-level analysis covers the major markets contributing to this forecast, with the key markets listed below.
Demand Outlook for Lung Cancer Therapeutics Market in India
India’s lung cancer therapeutics industry is projected to record a CAGR of 17.6% through 2036. India has a large lung cancer treatment base, leaving room for broader targeted therapy use as diagnosis and hospital oncology services improve. Demand for India lung cancer therapeutics is strongest in metro oncology networks and large private hospitals. Local access gains rely on affordability and molecular testing availability.
- Delhi-NCR cancer centers support targeted therapy demand through private oncology access and higher diagnostic test use.
- National Cancer Grid participation improves treatment standardization across tertiary centers and state cancer hospitals.
- Mumbai and Bengaluru oncology clusters enhance stronger access for biomarker-led therapies in private care settings.
Sales Analysis of Lung Cancer Therapeutics Market in China
The lung cancer therapeutics sector in China is expected to expand at a CAGR of 17.0% during the assessment period. China has a large lung cancer treatment base, giving the country strong volume potential for targeted therapies and immunotherapy across hospital oncology settings. Demand for China lung cancer therapeutics is shaped by hospital access and reimbursement review. Domestic and multinational suppliers must align pricing with formulary inclusion.
- Beijing and Shanghai oncology hospitals boost high-volume demand for mutation testing and targeted treatment access.
- National reimbursement negotiations influence access for imported oncology drugs across provincial hospital systems.
- Large urban hospital networks increase demand for small cell lung cancer treatment in advanced cases.
Demand Outlook for Lung Cancer Therapeutics Market in South Korea
South Korea’s lung cancer therapeutics industry is forecast to grow at a CAGR of 15.4% by 2036. South Korea’s meaningful lung cancer treatment base amplifies therapy demand across advanced cancer centers and specialist oncology networks. South Korea lung cancer therapeutics demand benefits from strong hospital systems and biomarker testing access. Therapy uptake is closely linked with reimbursement review and specialist prescribing.
- Seoul National University Hospital and Samsung Medical Center strengthen complex lung cancer treatment access.
- Korean National Cancer Center helps shape clinical pathways for high-burden cancers and precision oncology care.
- Seoul-based hospital networks create better access for EGFR and ALK-directed therapies as compared to smaller local systems.
Opportunity Analysis of Lung Cancer Therapeutics Market in Germany
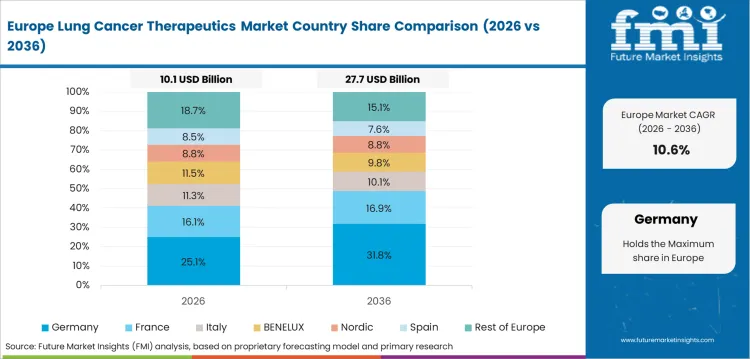
The lung cancer therapeutics segment in Germany is forecast to advance at 13.2% CAGR over the forecast period. Germany has a sizable lung cancer treatment base, and established reimbursement systems support demand for high-value oncology drugs after clinical review. Germany lung cancer therapeutics demand is strongest across university hospitals and specialist cancer centers. Suppliers need strong evidence packages for payer and hospital committee review.
- Berlin and Munich oncology centers increase demand for advanced NSCLC treatment and clinical trial-linked care.
- German statutory health insurance review affects branded oncology access across hospital and outpatient channels.
- University hospital networks sustain demand for immunotherapy and targeted regimens in molecularly selected patients.
Future Outlook for Lung Cancer Therapeutics Market in Australia
The Australia lung cancer therapeutics industry is expected to post 10.4% CAGR through 2036. Cancer Australia estimated 15,108 new lung cancer cases in Australia in 2025 and 8,994 deaths from lung cancer. Lung cancer remains a major treatment priority for national oncology planning. Australia lung cancer therapeutics demand gains from screening rollout and specialist access in major cities. Therapy volume will rely on earlier diagnosis and reimbursement listing quality.
- The National Lung Cancer Screening Program favors earlier case identification from July 2025.
- Sydney and Melbourne cancer centers strengthen access for targeted therapy and immunotherapy planning.
- Pharmaceutical Benefits Scheme listings influence branded therapy access across public and private oncology settings.
In-depth Analysis of Lung Cancer Therapeutics Market in the United States
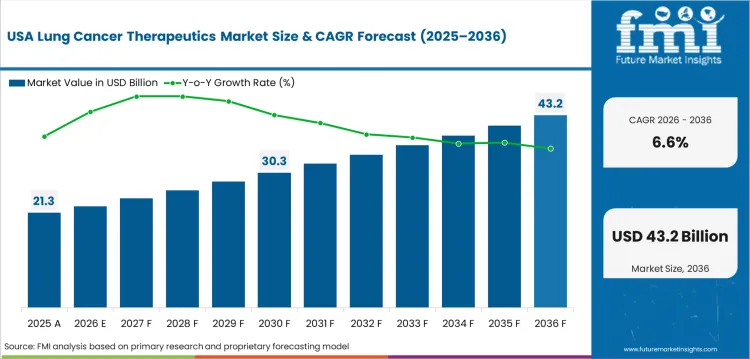
The USA lung cancer therapeutics industry is forecast to grow at 6.6% CAGR through 2036. American Cancer Society estimated 226,650 new lung cancer cases in the United States in 2025. The mature patient base keeps therapy demand large even with slower percentage expansion. USA lung cancer therapeutics demand is influenced by FDA approvals and payer coverage policies. NCCN aligned treatment pathways guide clinical practice across settings. Suppliers need trial depth and payer evidence to defend premium therapy access.
- FDA oncology approvals enhance rapid treatment pathway updates across major academic cancer centers.
- Medicare coverage decisions influence access for older lung cancer patients across community oncology practices.
- Texas and California oncology networks lead to large demand pools for cancer biological therapy use.
Sales Analysis of Lung Cancer Therapeutics Market in Japan
Japan’s lung cancer therapeutics sector is projected to record 8.4% CAGR through 2036. Japan has a large lung cancer treatment base, with steady therapy needs across older patient groups and specialist oncology settings. Japan lung cancer therapeutics demand is shaped by specialist oncology access and national reimbursement review. Manufacturers need clinical evidence suited to elderly treatment tolerance and mutation-defined selection.
- Tokyo and Osaka cancer centers raise demand for advanced NSCLC therapy and mutation testing access.
- Japan’s national health insurance system shapes branded oncology drug access after reimbursement review.
- High elderly cancer burden reinforces demand for tolerable small molecule and immunotherapy regimens.
Competitive Landscape and Strategic Positioning
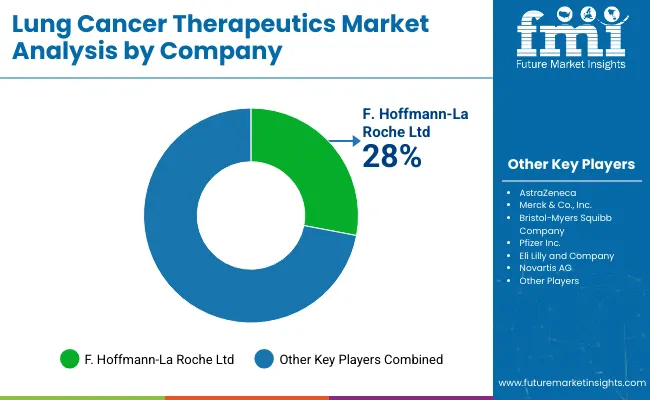
- AstraZeneca emphasizes deep EGFR therapy coverage and broad lung cancer treatment reach across major oncology markets.
- Merck & Co., Inc. and Bristol-Myers Squibb emphasize checkpoint inhibitor strength and approved immunotherapy regimens.
- Specialist oncology firms focus on mutation-defined assets for patient groups needing targeted treatment after biomarker confirmation.
Competition in the lung cancer therapeutics market depends on evidence depth, biomarker coverage, and oncology channel access. AstraZeneca holds a strong treatment position through Tagrisso and broader EGFR-mutated lung cancer treatment work across NSCLC care. Merck & Co., Inc. focuses on Keytruda across approved lung cancer treatment settings. Roche and Bristol-Myers Squibb provide hospital drugs with Tecentriq and Opdivo-based pathways.
Pfizer and Eli Lilly compete on kinase inhibitor assets within their specialty oncology portfolios. Novartis and Takeda serve targeted therapy demand through focused oncology portfolios. Boehringer Ingelheim and Amgen focus on selected mutation directed lung cancer assets. High development costs and strict evidence needs make branded oncology approvals difficult for smaller firms. Companies with stronger biomarker coverage and faster cancer center access planning are better placed to gain access. Checkpoint inhibitor depth strengthens suppliers active in PD-1 non-small cell lung cancer treatment across advanced NSCLC care.
Key Companies in the Lung Cancer Therapeutics Market
Global companies active in the lung cancer therapeutics market include:
- AstraZeneca, Merck & Co., Inc., and Roche have strong oncology infrastructure for targeted therapy, immunotherapy, and global access planning.
- Bristol-Myers Squibb, Pfizer, and Eli Lilly serve oncology teams through immune checkpoint therapy and small molecule treatment coverage.
- Novartis, Takeda, Boehringer Ingelheim, and Amgen hold specialist positions across mutation-specific agents and advanced oncology development.
Competitive Benchmarking: Lung Cancer Therapeutics Market
| Company | Biomarker Portfolio Depth | Immunotherapy Portfolio Readiness | Oncology Channel Reach | Geographic Footprint |
|---|---|---|---|---|
| AstraZeneca | High | High | Strong | Global |
| F. Hoffmann-La Roche Ltd | High | High | Strong | Global |
| Merck & Co., Inc. | High | High | Strong | Global |
| Bristol-Myers Squibb Company | High | High | Strong | Global |
| Pfizer Inc. | Medium | Medium | Strong | Global |
| Eli Lilly and Company | Medium | Medium | Strong | Global |
| Novartis AG | Medium | Medium | Strong | Global |
| Takeda Pharmaceutical Company Limited | High | Medium | Moderate | Global |
| Boehringer Ingelheim International GmbH | High | Medium | Moderate | Global |
| Amgen Inc. | Medium | Medium | Strong | Global |
Source: Future Market Insights, 2026.
Key Developments in Lung Cancer Therapeutics Market
- In December 2025, Johnson & Johnson announced USA FDA approval of RYBREVANT FASPRO™ (amivantamab and hyaluronidase-lpuj) for patients with EGFR-mutated non-small cell lung cancer. RYBREVANT FASPRO adds a subcutaneous administration option across approved RYBREVANT® indications and reduces treatment administration burden for EGFR-targeted therapy users.
- In December 2024, AstraZeneca received USA FDA approval for Imfinzi (durvalumab) for adults with limited-stage small cell lung cancer whose disease had not progressed after concurrent platinum-based chemotherapy and radiation therapy. The approval expands immunotherapy use into limited-stage SCLC after chemoradiotherapy.
Key Players in the Lung Cancer Therapeutics Market
Major Global Players:
- AstraZeneca
- F. Hoffmann-La Roche Ltd
- Merck & Co., Inc.
- Bristol-Myers Squibb Company
- Pfizer Inc.
- Eli Lilly and Company
- Novartis AG
Specialist Players:
- Takeda Pharmaceutical Company Limited
- Boehringer Ingelheim International GmbH
- Amgen Inc.
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 46.7 billion to USD 141.2 billion, at a CAGR of 11.7% |
| Market Definition | The lung cancer therapeutics market includes prescription drugs used to treat NSCLC and SCLC across chemotherapy, targeted therapy, angiogenesis, and immunotherapy regimens. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | India, China, South Korea, Germany, Australia, United States, Japan, and more than 30 countries |
| Key Companies Profiled | AstraZeneca, F. Hoffmann-La Roche Ltd, Merck & Co., Inc., Bristol-Myers Squibb Company, Pfizer Inc., Eli Lilly and Company, Novartis AG, Takeda Pharmaceutical Company Limited, Boehringer Ingelheim International GmbH, Amgen Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with treated patient pools and verified therapy use patterns. |
Source: Future Market Insights, 2026.
Lung Cancer Therapeutics Market by Segments
Lung Cancer Therapeutics Market Segmented by Drug Class:
- Cytotoxic Chemotherapy Drugs
- Targeted Therapies
- Angiogenesis Inhibitors
Lung Cancer Therapeutics Market Segmented by Formulation:
- Injectable
- Oral
Lung Cancer Therapeutics Market Segmented by Molecule Type:
- Small Molecule
- Large Molecule
Lung Cancer Therapeutics Market Segmented by Cancer Type:
- Non Small Cell Lung Cancer
- Small Cell Lung Cancer
Lung Cancer Therapeutics Market Segmented by Distribution Channel:
- Institutional Sales
- Retail Sales
Lung Cancer Therapeutics Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Europe
- Germany
- UK
- Italy
- Spain
- France
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Saudi Arabia
- Other GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Sources and Bibliography
- Canada’s Drug Agency. (2025, June). Pharmacoeconomic review. In Tarlatamab (Imdelltra): Therapeutic area: Extensive-stage small cell lung cancer: Reimbursement review. NCBI Bookshelf.
- Mudumba, R., Liu, X., Davis, I., Romley, J. A., & Nieva, J. J. (2025). Real-world costs, treatment patterns, and clinical outcomes associated with treatments for advanced anaplastic lymphoma kinase-positive non-small cell lung cancer. Journal of Managed Care & Specialty Pharmacy, 31(9), 890-899.
- Cancer Australia. (2026, February). Lung cancer in Australia statistics. Australian Government.
- American Cancer Society. (2025). Cancer Facts & Figures 2025. American Cancer Society.
- USA Food and Drug Administration. (2024, February). FDA approves osimertinib with chemotherapy for EGFR-mutated non-small cell lung cancer. USA Food and Drug Administration.
- USA Food and Drug Administration. (2025, June). FDA approves taletrectinib for ROS1-positive non-small cell lung cancer. USA Food and Drug Administration.
- USA Food and Drug Administration. (2024, September). FDA approves atezolizumab and hyaluronidase-tqjs for subcutaneous injection. USA Food and Drug Administration.
- USA Food and Drug Administration. (2024, October). FDA approves neoadjuvant/adjuvant nivolumab for resectable non-small cell lung cancer. USA Food and Drug Administration.
Bibliography is provided for reader reference and uses primary non-commercial government, regulatory, academic, or official health body publications.
This Report Answers
- What is the current and future size of the lung cancer therapeutics market?
- How fast is the lung cancer therapeutics market expected to expand between 2026 and 2036?
- Which drug class is expected to lead the market by 2026?
- Which formulation segment is expected to account for the most demand by 2026?
- What factors are driving demand for lung cancer therapeutics globally?
- How are biomarker evidence needs influencing adoption of targeted therapies?
- Why are institutional sales the main demand base for high-cost treatment regimens?
- How are advanced NSCLC pathways creating demand for specialized therapeutic portfolios?
- Which countries are projected to record faster expansion through 2036?
- Who are the key companies active in the lung cancer therapeutics market?
Frequently Asked Questions
What is the global market demand for Lung Cancer Therapeutics in 2026?
In 2026, the global market for lung cancer therapeutics is expected to be worth USD 46.7 billion.
How big will the market for Lung Cancer Therapeutics be in 2036?
By 2036, the market for lung cancer therapeutics is expected to be worth USD 141.2 billion.
How much is demand for Lung Cancer Therapeutics expected to expand between 2026 and 2036?
Between 2026 and 2036, demand for lung cancer therapeutics is expected to expand at a CAGR of 11.7%.
Which drug class segment is likely to be the leading drug class globally by 2026?
Targeted Therapies will make up 91.2% of the Drug Class segment in 2026.
Which formulation segment is expected to lead globally by 2026?
Injectable formulations are expected to account for 61.0% of formulation demand in 2026.
What is causing demand to rise in India?
The India lung cancer therapeutics market is projected to record 17.6% CAGR through 2036, backed by oncology access and diagnostic capacity.
What does this report mean by Lung Cancer Therapeutics Market definition?
The lung cancer therapeutics market includes prescription drugs used to treat NSCLC and SCLC across direct antitumor therapy pathways.
How does FMI make the Lung Cancer Therapeutics forecast and check it?
Forecasting models use a hybrid bottom-up and top-down approach, starting with therapy demand and checking it against oncology access patterns.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Tooling, Models, and Reference Databases
- Primary Modes
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Desk Research Programme (Secondary Evidence)
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Headlamps
- Chest lights
- Helmet Lights
- Handheld hybrids
- Headlamps
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Maintenance
- First responders
- Mining
- Warehousing
- Maintenance
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Safety Rating
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Safety Rating, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Safety Rating, 2026 to 2036
- General industrial
- Intrinsically safe
- Rugged waterproof
- General industrial
- Y to o to Y Growth Trend Analysis By Safety Rating, 2021 to 2025
- Absolute $ Opportunity Analysis By Safety Rating, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Li-ion Power
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Li-ion Power, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Li-ion Power, 2026 to 2036
- Rechargeable Li-ion
- Replaceable batteries
- Hybrid
- Rechargeable Li-ion
- Y to o to Y Growth Trend Analysis By Li-ion Power, 2021 to 2025
- Absolute $ Opportunity Analysis By Li-ion Power, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By End Use
- By Safety Rating
- By Li-ion Power
- Competition Analysis
- Competition Deep Dive
- Streamlight
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Pelican Products
- Wolf Safety
- Ledlenser
- Milwaukee Tool
- Klarus
- Energizer
- Eaton
- Bosch
- Streamlight
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Safety Rating, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Li-ion Power, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Safety Rating, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Li-ion Power, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Safety Rating, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Li-ion Power, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Safety Rating, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Li-ion Power, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Safety Rating, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Li-ion Power, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Safety Rating, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Li-ion Power, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Safety Rating, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Li-ion Power, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Safety Rating, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Li-ion Power, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value Share and BPS Analysis by Safety Rating, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Safety Rating, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Safety Rating
- Figure 12: Global Market Value Share and BPS Analysis by Li-ion Power, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Li-ion Power, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Li-ion Power
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Product Type
- Figure 29: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End Use
- Figure 32: North America Market Value Share and BPS Analysis by Safety Rating, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Safety Rating, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Safety Rating
- Figure 35: North America Market Value Share and BPS Analysis by Li-ion Power, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Li-ion Power, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Li-ion Power
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Product Type
- Figure 42: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by End Use
- Figure 45: Latin America Market Value Share and BPS Analysis by Safety Rating, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Safety Rating, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Safety Rating
- Figure 48: Latin America Market Value Share and BPS Analysis by Li-ion Power, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Li-ion Power, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Li-ion Power
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Product Type
- Figure 55: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by End Use
- Figure 58: Western Europe Market Value Share and BPS Analysis by Safety Rating, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Safety Rating, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Safety Rating
- Figure 61: Western Europe Market Value Share and BPS Analysis by Li-ion Power, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Li-ion Power, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Li-ion Power
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Safety Rating, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Safety Rating, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Safety Rating
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Li-ion Power, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Li-ion Power, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Li-ion Power
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Product Type
- Figure 81: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by End Use
- Figure 84: East Asia Market Value Share and BPS Analysis by Safety Rating, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Safety Rating, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Safety Rating
- Figure 87: East Asia Market Value Share and BPS Analysis by Li-ion Power, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Li-ion Power, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Li-ion Power
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Safety Rating, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Safety Rating, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Safety Rating
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Li-ion Power, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Li-ion Power, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Li-ion Power
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Safety Rating, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Safety Rating, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Safety Rating
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Li-ion Power, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Li-ion Power, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Li-ion Power
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

