Laparoscopic Devices Market
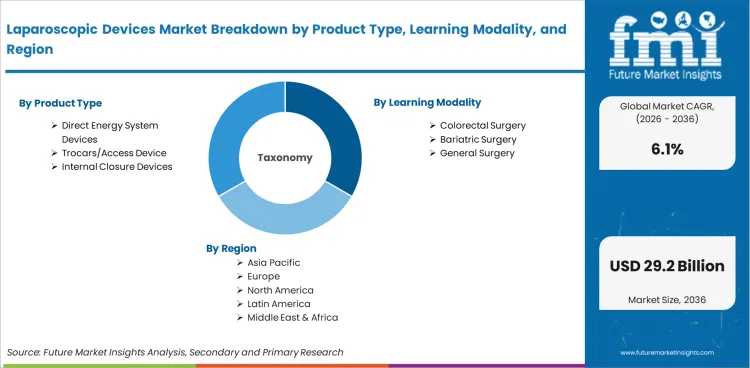
The laparoscopic devices market is segmented by Product Type (Direct Energy System Devices, Trocars/Access Devices, Internal Closure Devices, Laparoscopes, Hand Access Instruments, Insufflation Devices, Robotic Assisted Surgical Systems), Application (Bariatric Surgery, Colorectal Surgery, General Surgery, Gynecological Surgery, Other Laparoscopic Surgery), End User (Hospitals, Ambulatory Surgical Centers, Clinics), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Laparoscopic Devices Market size, market forecast and outlook By FMI
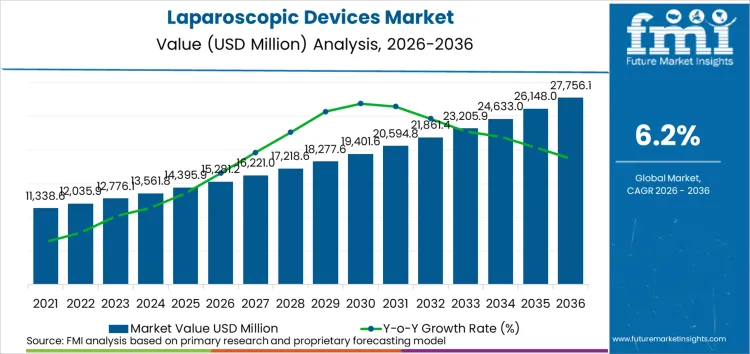
In 2026, the laparoscopic devices market is valued at USD 15,281.2 million. Based on the provided market model, demand is estimated to grow to USD 27,625.6 million by 2036. The market is projected to expand at a CAGR of 6.1% during the forecast period.
Absolute dollar growth of USD 12,344.4 million over the decade points to a market that is already well-established, yet still gaining procedure share across major surgical specialties. The underlying commercial logic is straightforward: laparoscopic surgery is not a single-product market. Each case pulls through access devices, visualization, insufflation, closure, and often-advanced energy. That creates a layered revenue model in which recurring procedural demand supports disposables and accessories, while capital equipment upgrades sustain the installed base.
Summary of Laparoscopic Devices Market
- Laparoscopic Devices Market Definition
- The industry covers the device ecosystem used in minimally invasive surgery, including access, visualization, insufflation, closure, energy delivery, and robotic assistance across abdominal and pelvic procedures.
- Demand Drivers in the Market
- Hospitals continue to shift appropriate cases toward minimally invasive approaches because shorter recovery and lower open-surgery burden remain clinically attractive.
- Large procedure categories such as colorectal and bariatric surgery keep device demand broad rather than concentrated in one specialty.
- Vendor portfolios that combine trocars, energy, visualization, and insufflation are better placed to retain accounts once an OR standard is set.
- Key Segments Analyzed in the FMI Report
- By Product Type: Direct Energy System Devices lead at 29.4% share in 2026, followed by Trocars/Access Devices at 20.5% share.
- By Application: Colorectal Surgery leads at 31.2% share in 2026, followed by Bariatric Surgery at 27.3% share.
- By End User: Hospitals lead at 72.6% share in 2026, followed by Ambulatory Surgical Centers at 19.0% share.
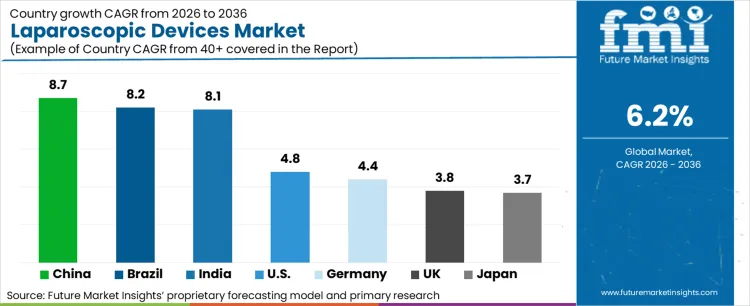
- Geography: China shows the fastest growth at 10.3% CAGR, followed by Brazil at 8.7% and India at 8.3%.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant at Future Market Insights, opines, “This market is controlled less by isolated product superiority and more by procedural fit. Once a hospital standardizes access, energy, optics, and service support, switching becomes harder unless there is a clear workflow or economic gain.”
- Strategic Implications and Executive Takeaways
- Build around platform depth, not isolated SKUs, because hospitals prefer fewer vendors across trocars, energy, visualization, and supporting accessories.
- Defend hospital accounts through surgeon training and OR compatibility, since device replacement is often constrained by established workflow and staff familiarity.
- Use robotics selectively as an account-expansion lever rather than a universal volume engine, because robotic adoption remains concentrated in capital-ready institutions. Intuitive’s da Vinci systems are positioned explicitly for hospitals seeking to increase minimally invasive surgery scale and efficiency, while Medtronic’s Hugo platform is being framed as a flexible OR-centric robotics investment.
- Methodology
- As per FMI, sizing and splits were reconciled using vendor portfolio mapping and procedure concentration across colorectal, bariatric, general, and gynecological surgery.
- Segment shares were validated by linking device demand to actual case flow, where each laparoscopic procedure typically pulls through multiple product categories rather than a single instrument class.
- Sensitivity checks were run across hospital and ambulatory site mix to confirm revenue movement under realistic shifts in procedural migration and capital refresh timing.
As John Langell, MD, highlighted the clinical relevance of Xenocor’s newly cleared Saberscope, “Every case begins with a new scope that delivers a clear, consistent image,” emphasizing that the single-use articulating laparoscope is intended to reduce contamination concerns, avoid reprocessing-related downtime, and help surgical teams stay focused on the procedure rather than equipment reliability. [1]
China (10.3% CAGR) is expected to be the fastest-growing tracked market, followed by Brazil (8.7%) and India (8.3%), where procedure volumes, hospital expansion, and improving access to minimally invasive surgery continue to widen the installed base. Japan (6.1%), Italy (5.8%), and Turkey (5.8%) sit in the next tier, where replacement demand and broader procedural adoption both matter. France (5.0%), the USA (4.6%), Germany (4.5%), and the UK (4.0%) remain commercially important, although growth there is shaped more by technology refresh, robotics layering, and account retention than by first-time market formation.
Laparoscopic Devices Market Definition
The market covers devices used to perform minimally invasive abdominal and pelvic surgery through small incisions using endoscopic visualization, specialized access ports, insufflation, tissue manipulation, closure, and energy delivery. It includes direct energy system devices, trocars and access devices, internal closure devices, laparoscopes, hand access instruments, insufflation devices, and robotic-assisted surgical systems used in laparoscopic workflows.
Commercial demand spans colorectal surgery, bariatric surgery, general surgery, gynecological surgery, and other laparoscopic procedures performed in hospitals, ambulatory surgical centers, and clinic-linked surgical settings. This is a procedure-driven equipment market, not a general hospital supply market.
Laparoscopic Devices Market Inclusions
The report includes global and regional market sizing and a forecast for 2026 to 2036. Product type, application, and end user, with country-level CAGR comparisons across key markets, provide segment-level sizing. Coverage includes revenue generated from laparoscopic capital equipment, reusable instruments, procedure-linked accessories, and disposable device categories used in minimally invasive surgical workflows.
The scope also captures how product choice is influenced by procedural complexity, OR standardization, smoke and visualization management, hospital procurement discipline, surgeon training, and robotics adoption in selected accounts. Official company product pages make clear that leading vendors do not compete in a single narrow category.
Laparoscopic Devices Market Exclusions
The scope excludes open-surgery-only instruments, non-laparoscopic diagnostic equipment, generic OR furniture, anesthesia systems, and post-operative care consumables that do not participate directly in laparoscopic procedure execution. It also excludes pharmaceuticals, sutures sold outside laparoscopic procedure sets, and broad imaging assets that are not integral to laparoscopic surgery workflows.
This distinction matters because procurement behavior in laparoscopy is procedure-linked and platform-led. A hospital may delay an imaging room refresh without materially changing laparoscopic case volume, but it cannot execute minimally invasive abdominal surgery without access devices, visualization, insufflation, and closure support fitted to that workflow. The commercial center of gravity remains the surgical stack and its case-by-case pull-through, not general hospital capital spending.
Laparoscopic Devices Market research methodology
- Primary research: Interviews were conducted with general surgeons, colorectal surgeons, bariatric specialists, gynecologic surgeons, OR managers, procurement teams, and supplier commercial personnel to validate product usage, account concentration, and replacement cycles.
- Desk research: Publicly available clinical guidance, hospital materials, surgical society publications, and company primary disclosures were reviewed to benchmark procedure pathways and vendor positioning.
- Market-sizing and forecasting: A hybrid model was applied using segment shares by product type, application, and end user, then reconciled through procedure intensity, installed-base logic, and country-level adoption patterns.
- Data validation and update cycle: Outputs were checked for internal consistency across segment splits, CAGR hierarchy, and the balance between disposable pull-through and capital equipment demand.
Segmental Analysis
Laparoscopic Devices Market Analysis by Product Type
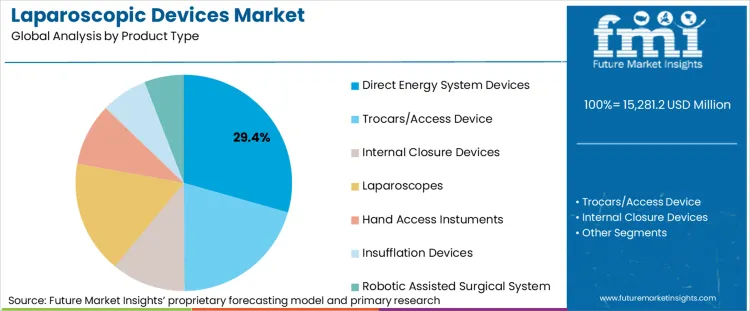
Based on the provided market model, Direct Energy System Devices are estimated to hold 29.4% share in 2026, followed by Trocars/Access Devices at 20.5%. Direct energy leads because energy delivery sits close to procedural criticality. Tissue dissection, sealing, and hemostasis influence operative speed, surgeon confidence, and case standardization more directly than many accessory categories. That makes energy harder to displace once surgeons and OR teams are trained on a given platform. Medtronic’s official portfolio presentation reflects this breadth, spanning electrosurgical products, vessel sealing, ultrasonic dissection, and related laparoscopic categories rather than treating energy as an add-on line.
- Workflow centrality: Energy devices are used during core procedural steps, which gives them greater clinical weight than supporting accessories. In October 2025, Olympus Corporation completed launch of THUNDERBEAT™ II for hemostatic dissection and large vessel transection1 in both laparoscopic and open surgery.[2]
- Training lock-in: Once surgeons become comfortable with activation feel, seal quality, and OR setup, switching becomes less attractive without a clear benefit.
- Portfolio protection: Vendors with strong energy franchises often cross-sell trocars, closure, and access products into the same account.
Laparoscopic Devices Market Analysis by Learning Modality
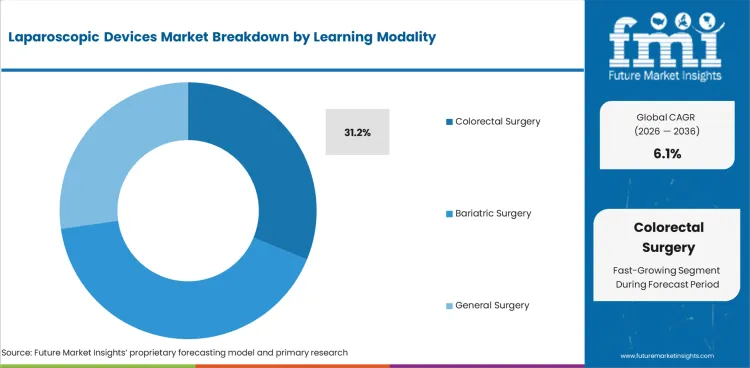
Colorectal Surgery accounts for 31.2% share in 2026, followed by Bariatric Surgery at 27.3%. Colorectal surgery leads because it combines substantial case volume with high device intensity. These procedures demand reliable access, stable pneumoperitoneum, clear visualization, dissection capability, tissue handling, and closure support. NCI continues to describe laparoscopic-assisted colectomy as a defined surgical approach in colon surgery, and Stryker’s insufflation materials explicitly position insufflation systems for colorectal and general surgery, underscoring the procedure’s commercial weight within laparoscopy. Bariatric surgery remains close behind because metabolic surgery still depends heavily on minimally invasive access, with NIDDK materials continuing to frame laparoscopic gastric procedures within mainstream bariatric practice.
- High device intensity: Colorectal cases typically require a broad mix of access, visualization, dissection, and closure tools. According to American Cancer Society (ACS) in 2026 alone, approximately 108,806 of new cases for colorectal cancer in US. [3]
- Institutional concentration: These procedures are often concentrated in hospitals and advanced surgical centers that buy on platform logic, not single-item price alone.
- Stable procedural base: Bariatric and colorectal surgery both keep laparoscopic demand linked to established clinical pathways rather than one-off experimental adoption.
Laparoscopic Devices Market Drivers, Restraints, and Opportunities
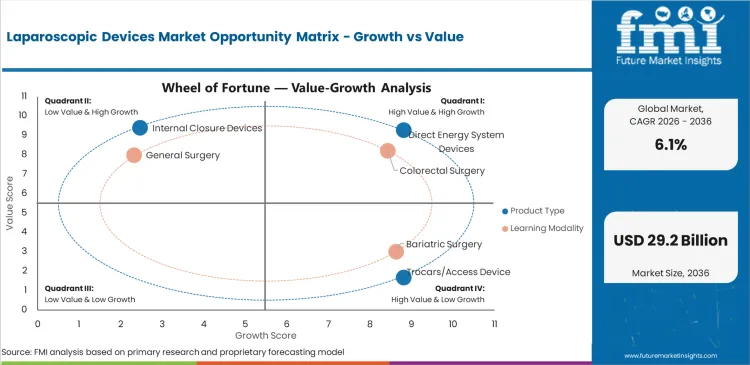
Future Market Insights analysis indicates that the market benefits from a durable clinical preference for minimally invasive surgery where appropriate, but growth is not automatic across all product lines. Procedural migration supports demand, yet buying decisions still pass through hospital committees, surgeon champions, and capital allocation cycles. That is why direct energy, optics, access, and insufflation often move together as platform decisions rather than as isolated purchases. SAGES and NHS materials support the clinical side of this logic, emphasizing recovery and morbidity advantages that keep laparoscopy structurally relevant.
The main restraint is not lack of procedural relevance. It is account complexity. Hospitals do not replace visualization systems, energy generators, or robotic platforms lightly once training, sterilization, service contracts, and surgeon familiarity are embedded. Robotics also adds a second layer of capital scrutiny. Intuitive’s da Vinci 5 rollout and Medtronic’s Hugo positioning show that the market is still moving toward more integrated and data-enabled OR environments, but this does not erase budget discipline. The opportunity lies with suppliers that reduce switching pain, fit existing OR workflows, and show practical value through efficiency, visualization quality, smoke control, or instrument versatility.
- Procedure migration driver: Minimally invasive surgery continues to gain procedural relevance because of recovery and morbidity advantages.
- Platform switching restraint: Standardization, training burden, and service contracts make supplier replacement slower than headline innovation suggests.
- Integrated workflow opportunity: Vendors that tie visualization, insufflation, energy, and robotics into a coherent OR proposition are better placed to expand share.
Regional Analysis
Based on the regional analysis, the laparoscopic devices market is segmented into North America, Latin America, Western Europe, East Asia, South Asia & Pacific, and Middle East & Africa across key surgical device markets. Regional performance is assessed using country-level demand signals linked to hospital capacity, minimally invasive surgery penetration, capital equipment access, procedural case mix, and supplier service reach.
.webp)
| Country | CAGR |
|---|---|
| USA | 4.6% |
| Brazil | 8.7% |
| UK | 4.0% |
| Germany | 4.5% |
| France | 5.0% |
| Italy | 5.8% |
| India | 8.3% |
| China | 10.3% |
| Japan | 6.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Laparoscopic Devices Market Analysis
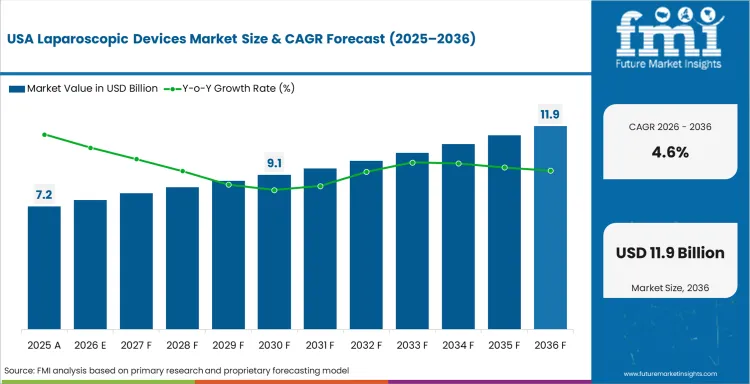
North America remains a high-value market because the installed base is deep, surgeon familiarity is high, and major multinationals have long-standing access to hospital procurement channels. Growth is steadier here than in faster-rising Asian markets, but the region still matters because capital refresh, robotic layering, and procedural intensity keep average account value high. The USA is not a volume story alone. It is also a mix story, where advanced imaging, energy platforms, and robotic systems sustain revenue quality.
- United States: The USA laparoscopic devices sales is expected to expand at 4.6% CAGR through 2036, supported by a increasing count of patient with colorectal cancer, sustained hospital capital spending in selected accounts, and ongoing replacement demand across visualization, access, and energy categories.
FMI’s report includes a detailed analysis of North America, along with a country-wise assessment that includes the USA, Canada, and Mexico. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
East Asia & South Asia Laparoscopic Devices Market Analysis
Asia is where procedure growth and infrastructure expansion are interacting most visibly. China and India benefit from expanding hospital capacity, broader access to specialist surgery, and a rising appetite for minimally invasive techniques in high-volume specialties. Japan remains commercially important for premium device categories, though its growth profile is more measured. Company product strategies also reflect Asia’s relevance. Olympus continues to emphasize advanced laparoscopic imaging, and Intuitive maintains dedicated regional positioning around robotic-assisted minimally invasive surgery.
- China: China is projected to rise at 10.3% CAGR through 2036, supported by fast hospital build-out, expanding adoption of minimally invasive procedures, and stronger uptake of advanced laparoscopic and robotic systems.
- India: India is set to grow at 8.3% CAGR through 2036, with momentum coming from widening surgical access, private hospital expansion, and stronger procurement of access, optics, and energy systems.
- Japan: Japan is expected to post 6.1% CAGR through 2036, with growth shaped by imaging quality, procedural precision, and technology upgrades rather than broad first-time adoption.
The full report analyzes the laparoscopic devices market across East and South Asia from 2026 to 2036, covering pricing, technology adoption, and growth drivers in China, Japan, India, and adjacent regional markets.
Latin America Laparoscopic Devices Market Analysis
Latin America remains distributor- and hospital-led, with Brazil acting as the principal anchor market. The region’s opportunity is not based on novelty alone. It rests on the widening use of minimally invasive surgery in tertiary centers and private hospital networks, where equipment availability, surgeon training, and after-sales support determine whether procedures scale beyond a handful of reference sites.
- Brazil: Brazil is forecast to grow at 8.7% CAGR through 2036, driven by wider procedural adoption in large hospitals, continued private-sector investment in minimally invasive surgery, and stronger device pull-through across colorectal, bariatric, and general surgery categories.
The report consists of a detailed analysis for the market in Brazil, Argentina, and Rest of Latin America. Readers can find detailed information about pricing analysis and regional trends affecting growth in the Latin America region.
Europe Laparoscopic Devices Market Analysis
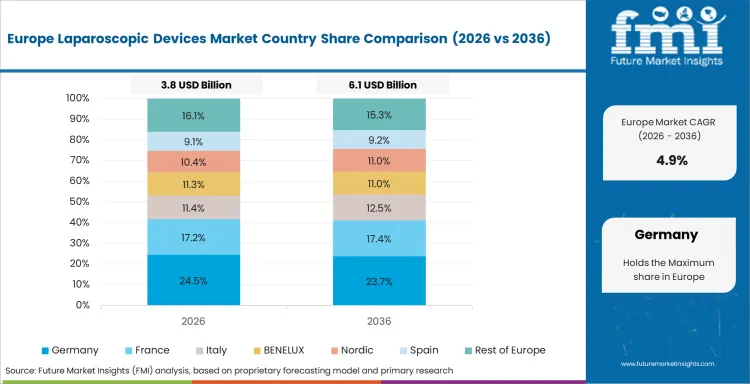
Europe remains a structurally important market because surgeon training, hospital standardization, and advanced visualization use are already well developed. Growth is moderate in the UK and Germany, firmer in Italy and France, and tied to replacement cycles, robotics layering, and imaging upgrades rather than wholesale market creation. Olympus’s 3D and 4K imaging positioning and Intuitive’s CE mark progress for da Vinci 5 illustrate how European accounts continue to be relevant for premium technology refresh.
- Germany: Germany is anticipated to expand at 4.5% CAGR through 2036, supported by mature minimally invasive surgery practice and recurring replacement demand in hospital systems.
- United Kingdom: The UK is projected to rise at 4.0% CAGR through 2036, with growth tied to capital discipline but continued use of laparoscopy across core specialties.
- France: France is expected to grow at 5.0% CAGR through 2036, with demand supported by established hospital-based MIS programs and imaging-led upgrades.
FMI’s analysis of the laparoscopic devices market in Europe consists of country-wise assessment that includes Germany, Italy, France, the United Kingdom, Spain, Nordic countries, Benelux, and Rest of Europe. Readers can know various regulations and latest trends in the regional market.
Competitive aligners for market players
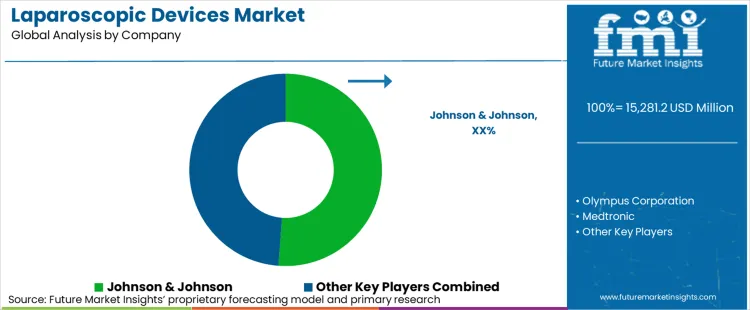
Market competition is fragmented on paper but concentrated in practice. A limited number of vendors have the breadth to compete credibly across energy, access, optics, insufflation, and robotics. That matters because hospitals do not buy laparoscopic devices the way they buy commodity consumables. They compare ecosystems. A trocar may be judged on unit price, but an account decision often depends on whether the same supplier can support the generator, visualization stack, service response, surgeon training, and OR consistency around that case type. Medtronic, Olympus, Stryker, and Intuitive each illustrate a different version of this ecosystem logic through their official portfolios.
Supplier retention is reinforced by workflow familiarity. Surgeons and OR staff get used to access feel, optical quality, energy behavior, smoke management, and service pathways. That makes switching slower than product announcements sometimes suggest. Incremental technology still matters, especially when it improves image clarity, reduces smoke-related disruption, or expands robotic workflow, but it usually has to fit existing procedural habits to win share. Olympus’s newer imaging platform positioning, Stryker’s insufflation workflow claims, and Intuitive’s da Vinci 5 launch all point in that direction: the message is not novelty for its own sake, but smoother execution inside minimally invasive surgery.
Buyer power remains meaningful because hospitals can stage evaluations, split categories by vendor, and use service expectations as a negotiation lever. Yet pricing is not the only trigger. In many accounts, switching happens only when there is a service gap, an unmet need in visualization or OR integration, or a capital refresh cycle that reopens the decision. That is why portfolio breadth acts as account protection. Vendors named in the current market universe include Janssen Pharmaceuticals, Inc. (Johnson & Johnson), Medtronic plc., Becton, Dickinson and Company, Olympus Corporation, B. Braun Melsungen AG, Stryker Corporation, Smith & Nephew Plc, KARL STORZ GmbH & Co. KG, Richard Wolf GmbH, CooperSurgical, Inc., Intuitive Surgical Inc, Endocontrol SA, Hangzhou Kangji Medical Instrument Co., Ltd, and Zhejiang Geyi Medical Instrument Co., Ltd.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In March 2026 the Olympus corp. launched ISERA ELITE™ III surgical imaging platform designed to enhance surgical visualization, workflow efficiency, and multispecialty flexibility in USA [4]
- In 2025, Intuitive received approval from CE for da Vinci 5 Surgical System in Europe for minimally invasive endoscopic procedures [5]
- In March 2025, LivsMed will unveil its ArtiSeal™ Vessel Sealing System at the SAGES conference, introducing the first 90° double-jointed articulating vessel sealer. Designed for enhanced surgical precision, the system enables perpendicular sealing with improved access and safety, signaling innovation in minimally invasive and laparoscopic surgical technologies.
Key Players in Laparoscopic Devices Market
- Janssen Pharmaceuticals, Inc. (Johnson & Johnson)
- Medtronic plc.
- Becton, Dickinson and Company
- Olympus Corporation
- B. Braun Melsungen AG
- Stryker Corporation
- Smith & Nephew Plc
- KARL STORZ GmbH & Co. KG
- Richard Wolf GmbH
- CooperSurgical, Inc.
- Intuitive Surgical Inc
- Endocontrol SA
- Hangzhou Kangji Medical Instrument Co., Ltd
- Zhejiang Geyi Medical Instrument Co., Ltd
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative units | US$ 15,287.2 million (2026) to US$ 27,625.6 million (2036), at a CAGR of 6.1% |
| Market definition | The market covers devices used to perform minimally invasive abdominal and pelvic surgery through small incisions using endoscopic visualization, specialized access ports, insufflation, tissue manipulation, closure, and energy delivery. It includes direct energy system devices, trocars and access devices, internal closure devices, laparoscopes, hand access instruments, insufflation devices, and robotic-assisted surgical systems used in laparoscopic workflows. Commercial demand spans colorectal surgery, bariatric surgery, general surgery, gynecological surgery, and other laparoscopic procedures performed in hospitals, ambulatory surgical centers, and clinic-linked surgical settings. This is a procedure-driven equipment market, not a general hospital supply market. |
| Product Type Segmentation | Direct Energy System Devices, Trocars/Access Device, Internal Closure Devices, Laparoscopes, Hand Access Instrument’s, Insufflation Devices, Robotic Assisted Surgical System |
| Application Segmentation | Bariatric Surgery, Colorectal Surgery, General Surgery, Gynecological Surgery, Other Laparoscopic Surgery |
| End User Coverage | Hospitals, Ambulatory Surgical Centers & Clinics |
| Regions covered | North America, Latin America, East Asia, South Asia, Western Europe, Eastern Europe, Middle East and Africa |
| Countries covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40 plus countries |
| Key companies profiled | Janssen Pharmaceuticals, Inc. (Johnson & Johnson), Medtronic plc., Becton, Dickinson and Company, Olympus Corporation, B. Braun Melsungen AG, Stryker Corporation, Smith & Nephew Plc, KARL STORZ GmbH & Co. KG, Richard Wolf GmbH, CooperSurgical, Inc., Intuitive Surgical Inc, Endocontrol SA, Hangzhou Kangji Medical Instrument Co., Ltd, Zhejiang Geyi Medical Instrument Co., Ltd |
| Forecast period | 2026 to 2036 |
| Approach | A hybrid top-down and bottom-up market modeling approach was used, validated through primary interviews with laparoscopic surgeons, operating room managers, hospital procurement teams, and device manufacturers, and further supported by product portfolio mapping, procedure-volume assessment, and end-user channel benchmarking, as per FMI. |
Laparoscopic Devices Market Analysis by Segments
Product Type
- Direct Energy System Devices
- Trocars/Access Device
- Internal Closure Devices
- Laparoscopes
- Hand Access Instrument’s
- Insufflation Devices
- Robotic Assisted Surgical System
Application
- Bariatric Surgery
- Colorectal Surgery
- General Surgery
- Gynecological Surgery
- Other Laparoscopic Surgery
End User
- Hospitals
- Ambulatory Surgical Centers
- Clinics
Region
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- Western Europe
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- Eastern Europe
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- Middle East & Africa
- Saudi Arabia
- Türkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] PR Newswire. Xenocor Announces FDA Clearance of Saberscope®, the First Single-Use 5mm Articulating Laparoscope for Enhanced Surgical Visualization. PR Newswire.
- [2] Olympus. Olympus Launches New Surgical Energy Device for Hemostatic Cutting and Vessel Sealing. Olympus News.
- [3] Cancer Research Institute. Colorectal Cancer Rates Are Skyrocketing in Young Adults - Is Your Lifestyle Putting You at Risk? Cancer Research Institute Blog.
- [4] Olympus. Olympus Launches Next-Generation Surgical Imaging Platform in USA Olympus News.
- [5] Intuitive Surgical. Intuitive’s da Vinci 5 Surgical System Receives CE Mark. Intuitive Surgical News Release.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This report addresses
- Market intelligence to support structured strategic decision making across minimally invasive surgery adoption, device positioning, and hospital-based procedural expansion strategies
- Market size estimation and 10-year revenue forecasts from 2026 to 2036 for laparoscopic devices demand across core surgical applications
- Growth opportunity mapping across product type, application, end user, and country-level demand pools
- Segment and regional revenue forecasts aligned to the stated taxonomy and covered geographies
- Competition strategy assessment including portfolio differentiation, operating room workflow fit, service support strength, and channel coverage benchmarking
- Product and procedure tracking focused on direct energy systems, access devices, laparoscopes, insufflation systems, closure devices, and robotic-assisted surgical platforms
- Regulatory and surgical practice environment impact analysis covering minimally invasive surgery standards, product handling requirements, sterilization considerations, and provider-use conditions
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for laparoscopic devices in the global market in 2026?
Demand for laparoscopic devices in the global market is estimated to be valued at US$ 15,281.2 Mn in 2026, as per FMI.
What will be the market size of laparoscopic devices in the global market by 2036?
Market size for laparoscopic devices is projected to reach US$ 27,625.6 Mn by 2036.
What is the expected demand growth for laparoscopic devices in the global market between 2026 and 2036?
Demand for laparoscopic devices in the global market is expected to grow at a CAGR of 6.1% between 2026 and 2036.
Which product type is poised to lead global demand by 2026?
Direct energy system devices solutions kits is expected to be the dominant product, capturing 29.4% share in 2026.
Which application is expected to account for the largest share in 2026?
Colorectal surgery is expected to hold the highest share at 31.2% in 2026.
Which end user is expected to dominate the mix in 2026?
Hospitals is expected to lead material use with 72.6% share in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Direct Energy System Devices
- Trocars/Access Device
- Internal Closure Devices
- Direct Energy System Devices
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Learning Modality
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Learning Modality, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Learning Modality, 2026 to 2036
- Colorectal Surgery
- Bariatric Surgery
- General Surgery
- Colorectal Surgery
- Y to o to Y Growth Trend Analysis By Learning Modality, 2021 to 2025
- Absolute $ Opportunity Analysis By Learning Modality, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Learning Modality
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Learning Modality
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Learning Modality
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Learning Modality
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Learning Modality
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Learning Modality
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Learning Modality
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Learning Modality
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Learning Modality
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Learning Modality
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Learning Modality
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Learning Modality
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Learning Modality
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Learning Modality
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Learning Modality
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Learning Modality
- Competition Analysis
- Competition Deep Dive
- Janssen Pharmaceuticals, Inc. (Johnson & Johnson)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Medtronic plc.
- Becton, Dickinson and Company
- Olympus Corporation
- B. Braun Melsungen AG
- Stryker Corporation
- Janssen Pharmaceuticals, Inc. (Johnson & Johnson)
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Learning Modality, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Learning Modality, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Learning Modality, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Learning Modality, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Learning Modality, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Learning Modality, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Learning Modality, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Learning Modality, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Learning Modality, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Learning Modality, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Learning Modality
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Learning Modality, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Learning Modality, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Learning Modality
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Learning Modality, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Learning Modality, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Learning Modality
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Learning Modality, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Learning Modality, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Learning Modality
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Learning Modality, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Learning Modality, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Learning Modality
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Learning Modality, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Learning Modality, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Learning Modality
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Learning Modality, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Learning Modality, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Learning Modality
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Learning Modality, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Learning Modality, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Learning Modality
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

