Dietary Supplement Market
The dietary supplements market is segmented by Customer Orientation (Women, Men, Senior Citizen, Toddlers/Kids), Primary Ingredients (Vitamins, Minerals, Omega 3, Botanicals Herbs or Compounds, Prebiotics & Probiotics, Collagen, Amino Acid, Enzymes, Protein), Form (Tablets & Capsules, Gummies & Chews, Powder, Soft Gel, Liquid, Others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Dietary Supplements Market Size, Market Forecast and Outlook By FMI
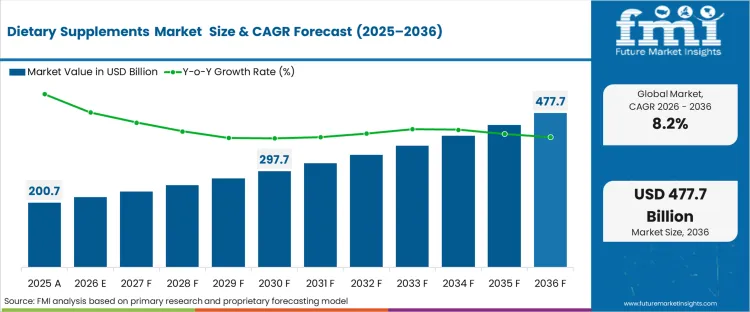
The dietary supplements market crossed a valuation of USD 200.9 billion in 2025. The industry is projected to reach USD 217.2 billion in 2026 at a CAGR of 8.2% during the forecast period. Demand outlook lifts the market valuation to USD 478.7 billion by 2036 as expanding nutraceutical innovation and digital health awareness strengthen demand for targeted supplementation across global populations.
Buyers are moving away from generic multivitamin routines and choosing stackable products built around specific health goals. This shift is pushing formulators in the preventive healthcare supplements market to define a clearer role for each product within the daily regimen. Brands that delay this shift may lose share to focused players built around distinct metabolic, immune, or lifestyle needs. Broad-spectrum positioning is becoming less effective as more consumers make supplement choices through diagnostic and outcome-based thinking. Commercial success now depends more on linking specific dietary supplements with measurable wellness results.
Summary of Dietary Supplements Market
- Dietary Supplements Market Definition
- This sector structurally defines the commercialization of concentrated physiological compounds in dose form. It bridges the gap between baseline dietary intake and optimized metabolic function, governed by distinct regulatory frameworks that separate it from conventional food matrices.
- Demand Drivers in the Market
- Dietary supplement market trends 2026 point to rising diagnostic accessibility compelling primary care practitioners to recommend targeted micronutrient interventions.
- Shifting insurance incentive models force corporate wellness directors to subsidize preventative supplementation programs.
- Declining soil nutrient density pushes consumer nutritionists to integrate exogenous baselines into standard dietary protocols.
- Key Segments Analyzed in the FMI Report
- Form: Tablets & Capsules are set to hold 64.8% share in 2026, as this form delivers maximum active ingredient payload per serving while ensuring superior shelf-stability for moisture-sensitive APIs.
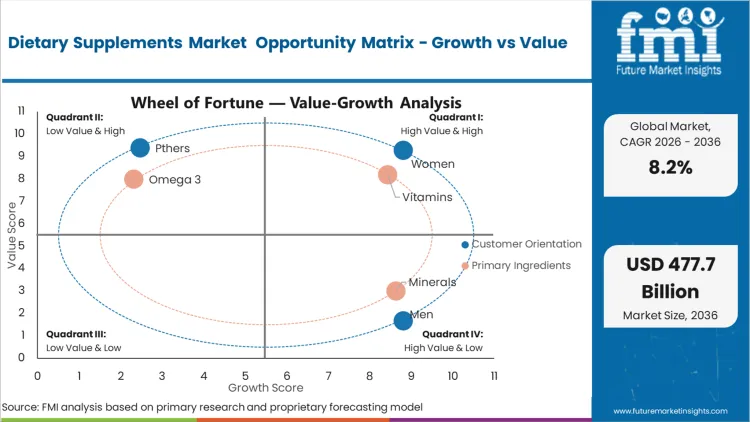
- Primary Ingredients: Vitamins are anticipated to record 33.3% share in 2026 within the vitamins and supplements market, driven by universally established baseline deficiency protocols across both developing and mature healthcare networks.
- Customer Orientations: Women are likely to grab 18.9% share in 2026, structurally necessitated by specific physiological cycles spanning reproductive health and bone density preservation.
- India: 10.5% compound growth, driven by digital-first distribution platforms bypassing traditional physical pharmacy gatekeepers.
- Analyst Opinion at FMI
- Nandini Roy Choudhury, Principal Analyst, Food and Beverage, at FMI, opines, "Many executives believe that clinical efficacy alone secures recurring revenue. In reality, a technically perfect dose provides zero commercial value if the patient abandons it due to pill fatigue. Contract manufacturers that master complex suspension matrices are capturing market share because they solve the compliance problem through specialized nutraceutical gummies, overriding the traditional preference for high-payload capsules."
- Strategic Implications / Executive Takeaways
- Formulation directors must prioritize bioavailability validation to defend premium pricing tiers against commoditized synthetic alternatives.
- Retail category managers should reconfigure shelf space away from generic multivitamins to avoid stagnation in single-digit growth categories.
- Contract manufacturers face margin compression that requires immediate investment into high-throughput chewable and liquid suspension infrastructure.
- Methodology
- Primary Research: Direct interviews with specialized nutrition buyers and contract formulation leads.
- Desk Research: Aggregation of verified ingredient import logs and clinical validation registries.
- Market-Sizing and Forecasting: Built upon raw material consumption rates and aligned with retail distribution volumes.
- Data Validation and Update Cycle: Triangulated against independent demographic health audits and continuously updated through proprietary algorithmic baseline adjustments.
Further growth in the dietary supplements market will depend on stronger links between personalized dosing and continuous biomarker tracking. Diagnostic platforms are helping drive this change by moving consumers away from symptom-based trial and error toward more data-backed supplement routines. When immune health supplements and metabolic support products are aligned more closely with recurring blood panel data, adherence is likely to improve and repeat purchasing may become more stable.
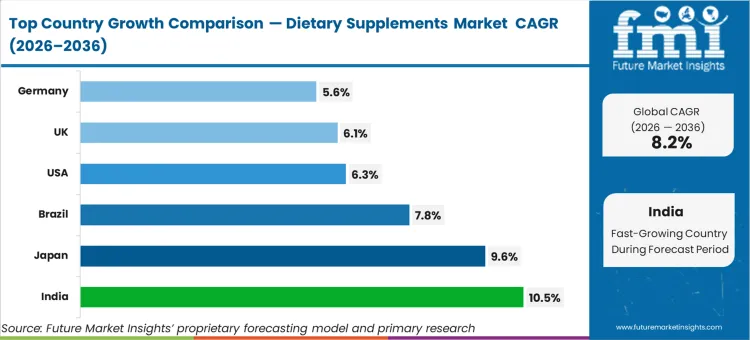
India is poised to expand at a 10.5% CAGR as rapid penetration of organized direct-to-consumer nutraceutical retail bypasses traditional pharmacy bottlenecks. Japan follows, advancing at 9.6% driven by an aging demographic structurally dependent on preventative longevity formulations. Brazil tracks at a 7.8% compound rate, while the USA is expected to register a 6.3% pace. The UK and Germany are estimated to grow at 6.1% and 5.6% respectively. The divergence across these territories within the dietary supplements market forecast reflects a fundamental transition from acute deficiency correction to baseline longevity optimization.
Supply Chain Analysis in the Dietary Supplements Market
Value creation in this supply chain shifts steadily from basic raw material handling toward formulation, downstream processing, and channel access. At the front end, raw material producers and primary processors form the supply base, supplying inputs either to domestic formulation plants or into export channels, which shows that upstream activity is relatively fragmented and volume-driven. The middle of the chain is where commercial leverage becomes clearer, manufacturing and processing plants convert raw inputs into higher-value dietary ingredient formats, while international processing links expand supply flexibility and create a parallel route for cross-border trade. Downstream, the structure becomes more market-facing, with processors, traders, importers, suppliers, and distributors acting as the bridge between ingredient production and final retail movement. That makes the later stages especially important for margin capture, product specification, and market reach, since access to end-use companies, export networks, and retail channels appears to determine how efficiently products move from ingredient stage to consumer purchase. Overall, the diagram suggests a supply chain that is not simply linear, but commercially tiered, with the strongest strategic control sitting in processing, specification, and distribution rather than in raw material origination alone.
Dietary Supplements Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 217.2 billion |
| Industry Value (2036) | USD 478.7 billion |
| CAGR (2026 to 2036) | 8.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Dietary Supplements Market Definition
The sector encompasses concentrated sources of nutrients or other substances with a nutritional or physiological effect, delivered in dose form. It is functionally distinguished from functional foods by its precise dosing architecture and extraction methods. The boundary defining this space is its reliance on non-prescription health optimization rather than acute disease treatment.
Dietary Supplements Market Inclusions
Scope inclusions cover concentrated nutrient delivery systems designed to supplement the normal diet. This involves single-ingredient and complex formulations spanning vitamins, minerals, amino acids, enzymes, and specialized omega 3 fatty acid extractions. Botanical extracts, performance proteins, and targeted probiotic strains formatted into capsules, powders, chewables, and liquids for direct consumer administration are fully integrated into the analysis.
Dietary Supplements Market Exclusions
The analysis explicitly excludes functional foods, fortified beverages, and meal replacement products. Their primary functional role remains caloric sustenance and hydration, requiring different regulatory compliance and retail distribution channels. Prescription-only clinical nutrition and parenteral feeding solutions are also excluded because their procurement is dictated by medical necessity rather than consumer health decisions.
Dietary Supplements Market Research Methodology
- Primary Research: Procurement directors at national pharmacy chains, formulation scientists at contract manufacturing organizations, and regulatory compliance officers guiding nutraceutical approvals.
- Desk Research: National dietary intervention registries, centralized adverse event reporting databases, and verified clinical trial endpoints for specific active pharmaceutical ingredients.
- Market-Sizing and Forecasting: Baseline sizing anchors to raw material extraction volumes and active ingredient import data, triangulated against finished product retail scan data.
- Data Validation and Update Cycle: Forecasts are cross-validated against demographic aging models and chronic disease prevalence shifts on an annualized review cycle.
Segmental Analysis
Dietary Supplements Market Analysis by Customer Orientation
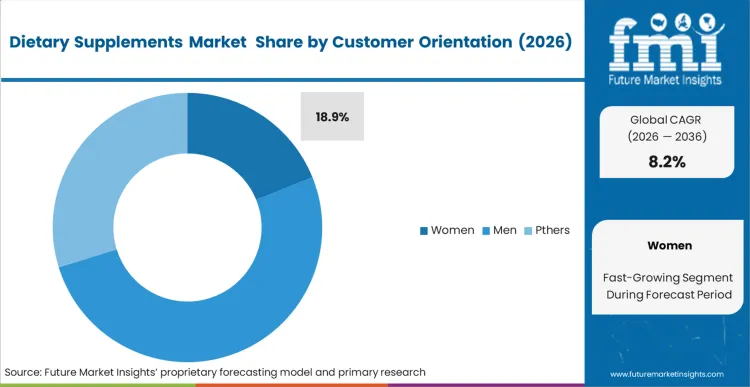
Targeted intervention across key physiological transitions is a major reason the women segment remains important in this market. Adoption is often linked to specific lifecycle needs, including prenatal support and post-menopausal bone health, rather than broad wellness positioning alone. This demand pattern places the Women segment at an 18.9% share. FMI analysis suggests that the inclusion of specialized sports nutrition in female recovery routines is also widening the application base. When formulations do not reflect these endocrine and lifecycle differences, regimen continuation tends to weaken. Brands that continue using unisex dosing strategies may face lower acceptance in retail environments that increasingly favor targeted efficacy. The personalized nutrition supplements market responds to this shift by aligning formulations more closely with individual hormone profiles.
- Bioavailability Failure: Standard formulations often fail to cross the absorption threshold during specific hormonal transitions. Consumers abandon the regimen entirely when blood markers show no physiological improvement.
- Payload Limitations: Combining necessary calcium and iron doses into a single daily matrix creates competitive absorption risks. Formulators must split doses operationally to achieve the intended clinical endpoint.
- Format Fatigue: Continuous daily pill requirements lead to sporadic compliance over long-term prevention cycles. Switching to high-concentration chewables allows users to maintain steady-state serum levels without resistance.
Dietary Supplements Market Analysis by Primary Ingredients
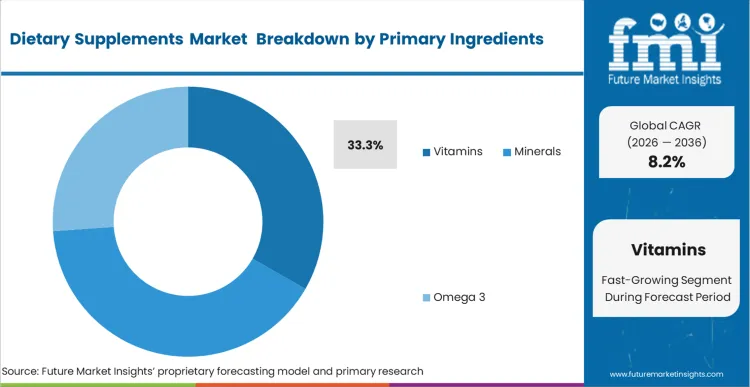
Rising concern around inadequate baseline nutrition from diet alone is increasing the shift toward concentrated supplementation. This demand pattern places Vitamins at a 33.3% share of the market, supported by broad deficiency concerns and their growing role in routine care recommendations. FMI analysis suggests that brand positioning in this segment is increasingly influenced by the choice between synthetic and whole-food-derived formats. Premium-tier players often strengthen this positioning by pairing vitamins with more complex protein formulations to support higher-value offerings. Brands that continue relying on older synthetic isolates may face greater commoditization as buyers become more informed and selective.
- Initial Trigger: Documented blood panel deficiencies force the initial consumer purchase to correct acute physiological imbalances.
- Qualification Standard: Buyers validate long-term choices by verifying third-party testing certifications and bioavailability claims for specific vitamin and mineral supplement products.
- Expansion Criteria: Once baseline trust is established, consumers expand their stack to include adaptogens and herbal supplements from the same qualified vendor.
Dietary Supplements Market Analysis by Form
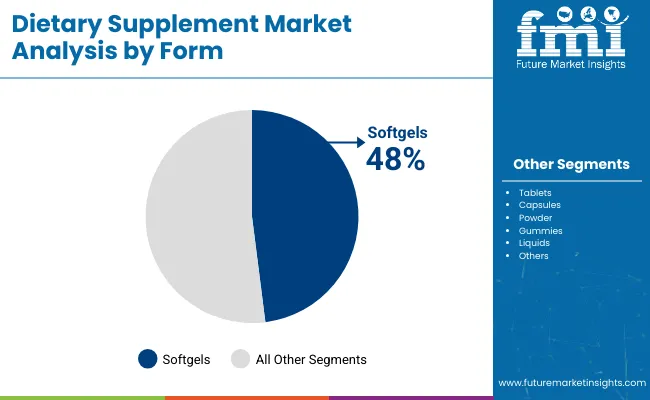
Tablets & Capsules account for 64.8% of the sector because they provide the only architecture capable of shielding volatile moisture-sensitive actives while maximizing raw payload. According to FMI's estimates, this operational necessity outweighs the consumer preference for novel formats in clinical-grade dosages. Advanced gummy supplements force traditional encapsulates to innovate their coating technologies. Dietary supplement contract manufacturing facilities that fail to optimize high-speed encapsulation lines while simultaneously developing vitamin gummies face severe capacity underutilization.
- Procurement Savings: Standardized hard-shell encapsulation utilizes legacy high-speed machinery. This generates massive cost efficiencies during initial production runs for dietary supplement suppliers.
- Hidden Expenditures: Moisture protection and specialized blistering add secondary packaging costs that erode baseline margins during the logistics phase.
- Lifecycle Comparison: When evaluating total stability over a two-year shelf life, standard capsules vastly outperform liquid suspensions, preserving the active ingredient claim until the end user consumes it.
Dietary Supplements Market Drivers, Restraints, and Opportunities
When evaluating what is driving the dietary supplements market, the structural pressure of predictive healthcare diagnostics stands out. This technology compels primary care practitioners to embed exogenous supplementation directly into preventative treatment plans. Diagnostic data allows practitioners to mandate high-concentration omega 3 concentrates before cardiovascular risk parameters peak. The fundamental commercial dynamic shifts from a discretionary consumer choice to a biologically necessitated recurring purchase. Formulators must adapt to this prescriptive model to capture long-term subscription loyalty, as generalist brands competing on broad awareness simply cannot match the retention rates of data-validated regimens.
Internal qualification cycles and bioavailability validation present the single biggest structural friction for market expansion. Consumers and practitioners frequently halt regimens because standard formulations fail to register significant shifts in actual physiological baselines. This clinical delivery failure is especially prominent in volatile herbal nutraceuticals where standard extracts lose potency during digestion. Liposomal delivery systems are emerging as a partial solution. Their high manufacturing overhead restricts them to the premium tier, leaving the mass-market segment vulnerable to high abandonment rates.
Opportunities in the Dietary Supplements Market
- Diagnostic Integration: Wearable technology algorithms identifying specific recovery deficits enable continuous adjustment. Specialized direct-to-consumer brands can capture this by linking their fulfillment systems directly to third-party health API data, revealing massive dietary supplements market opportunities.
- Substrate Engineering: Advancements in repulpable blister packaging allow sustainable material transition without compromising moisture barriers. Operations directors capturing this capability bypass upcoming extended producer responsibility penalties.
- Microbiome Personalization: Strain-specific sequencing data enables the deployment of highly customized probiotic supplements. A dedicated private label dietary supplements supplier can capture this by developing modular blending lines capable of short-run production.
Regional Analysis
Based on the regional analysis, the dietary supplements market is segmented into Americas, Europe, and Asia Pacific across 40+ countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| USA | 6.3% |
| UK | 6.1% |
| Germany | 5.6% |
| India | 10.5% |
| Brazil | 7.8% |
| Japan | 9.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Americas Dietary Supplements Market Analysis
The regulatory environment governing health claims determines exactly how formulators deploy capital in this territory. In FMI's view, the reliance on structural-function claims rather than strict pharmaceutical endpoints allows for rapid product iteration. It places the burden of proof entirely on the consumer's perceived efficacy. This policy framework accelerates the speed to market for novel ingredient combinations. Companies operating here compete heavily on format innovation and immediate shelf appeal to capture transient consumer interest.
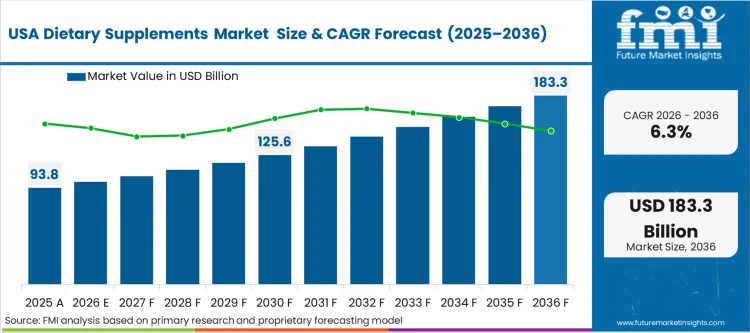
- USA: The regulatory distinction allowing structure-function claims fundamentally accelerates how quickly new North American dietary supplements hit retail shelves in the dietary supplements market USA. Procurement directors must filter thousands of SKUs and rely heavily on third-party certifications. The market is forecast to register a CAGR of 6.3%. Companies that integrate biomarker diagnostics into their retail platforms capture long-term subscription contracts rather than one-off purchases.
- Brazil: The complex network of import tariffs forces international brands to establish localized formulation partnerships rather than importing finished goods. Supply chain managers in Brazil must prioritize raw ingredient sourcing to bypass crippling logistics delays. Brazil's industry is expected to demonstrate a CAGR of 7.8%. The compound rate reflects the operational reality of navigating fragmented import frameworks.
FMI's report includes extensive coverage of surrounding Latin American and Canadian territories. The structural pattern across these additional countries reveals a unified pivot toward nearshoring raw botanical extraction to avoid cross-border regulatory friction.
Europe Dietary Supplements Market Analysis
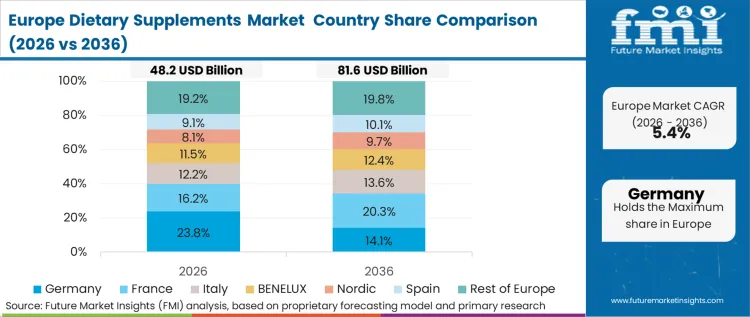
Stringent qualification standards and deeply embedded procurement practices shape the behavioral environment across this region. Buyers here demand complete supply chain transparency and verifiable clean-label certifications before authorizing shelf placement. Based on FMI's assessment, this buyer behavior-led environment suppresses the rapid proliferation of fad ingredients. Established players with the capital to maintain rigorous quality control frameworks secure the majority of retail contracts.
- Germany: Strict adherence to centralized European Food Safety Authority directives forces formulators to validate every health claim through rigorous clinical dossiers. Regulatory compliance officers operating here focus their efforts on substantiating evidence rather than rapid prototyping. The German sector is poised to expand at a 5.6% CAGR. Formulators operating here establish a compliance baseline that automatically qualifies their products for adjacent EU territories.
- UK: Retail procurement practices heavily favor brands that can provide immediate digital traceability of raw material origins. Compliance teams must integrate verifiable digital ledgers to pass initial vendor screening. A CAGR of 6.1% is expected for the UK over the forecast period. Retail buyers reduce their qualification cycles for new SKUs when manufacturers provide complete traceability data.
FMI's report includes a comprehensive assessment of the broader dietary supplements demand across the continent. Regional procurement networks consistently penalize brands that attempt to bypass standardized clean-label parameters.
Asia Pacific Dietary Supplements Market Analysis
When analyzing which countries are growing fastest in dietary supplements, the physical and digital infrastructure condition fundamentally shapes the adoption curve across these markets. Traditional pharmacy networks are being rapidly bypassed by aggressive direct-to-consumer digital platforms. As per FMI's projection, this infrastructure-led shift removes the historical bottleneck of limited physical shelf space. Highly specialized niche formulations can now reach fragmented consumer bases directly.
- India: The rapid deployment of specialized digital-first nutraceutical platforms entirely circumvents the disorganized traditional pharmacy network in the dietary supplements market India. Digital supply chain directors operate under conditions where last-mile delivery algorithms dictate market penetration. FMI estimates the Indian sector to expand at an annual growth rate of 10.5%. The advance signals a permanent pivot toward digital-first distribution.
- Japan: Advanced localized manufacturing infrastructure explicitly caters to an aging demographic. Product developers focus heavily on liquid suspensions and rapid-dissolve matrices suitable for reduced digestive capacity. Japan is expected to see its sector grow at a compound annual rate of 9.6%. Aging populations shift the baseline from acute recovery to preventative longevity.
FMI's report includes deep dive analytics on emerging Southeast Asian and Oceanic territories, as well as the dietary supplements market China. Across these additional countries, infrastructure investments are heavily skewed toward localized gummy and soft-gel encapsulation facilities to counter high import costs.
Competitive Aligners for Market Players
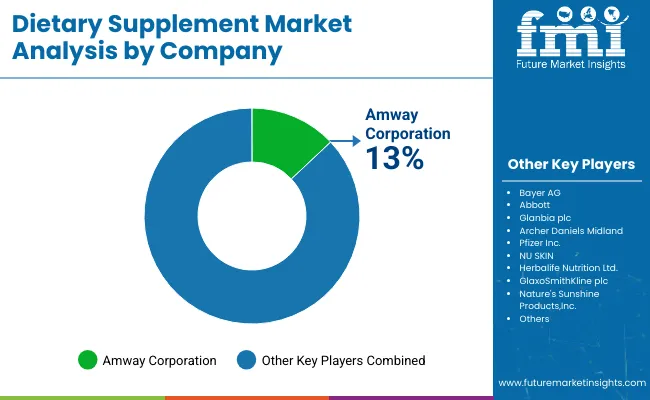
The Dietary Supplements Market is led by a group of established companies with strong brand recognition, broad product coverage, and wide market reach. Bayer AG and Nestlé Health Science benefit from global scale and diversified wellness portfolios. Amway and Herbalife continue to hold strong positions through direct selling models and brand-led nutrition programs. NOW Foods and Pharmavite are more closely connected with the core supplements category, supported by broad portfolios in vitamins, minerals, and targeted health products. Unilever has built relevance in the space through wellness-focused brands such as OLLY and SmartyPants, showing how large consumer companies are strengthening their supplements presence through brand development and acquisitions. Competition in this market depends heavily on consumer trust, product relevance, and the ability to stay visible across retail, e-commerce, practitioner-led channels, and direct-to-consumer platforms.
Key Players in Dietary Supplements Market
- Bayer AG
- Nestlé Health Science
- Amway
- Herbalife
- NOW Foods
- Pharmavite
- Unilever
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 217.2 billion to USD 478.7 billion, at a CAGR of 8.2% |
| Market Definition | The sector encompasses concentrated physiological compounds delivered in precise dose forms to optimize human metabolic baselines. It provides a structural bridge between conventional dietary intake and clinical pharmaceutical intervention. |
| Customer Orientation Segmentation | Women, Men, Senior Citizen, Toddlers/Kids |
| Primary Ingredients Segmentation | Vitamins, Minerals, Omega 3, Botanicals Herbs or Compounds, Prebiotics & Probiotics, Collagen, Amino Acid, Enzymes, Protein (Whey, Casein) |
| Form Segmentation | Tablets & Capsules, Gummies & Chews, Powder, Soft Gel, Liquid, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | USA, UK, Germany, India, Brazil, Japan, and 40 plus countries |
| Key Companies Profiled | Bayer AG, Nestlé Health Science, Amway, Herbalife, NOW Foods, Pharmavite, Unilever |
| Forecast Period | 2026 to 2036 |
| Approach | Formulation leads and procurement officers were interviewed to establish structural demand baselines. Baseline sizing anchors to active ingredient import volumes and raw material extraction data. Forecasts were cross-validated using chronic disease prevalence shifts and annualized retail audit registries. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Dietary Supplements Market Analysis by Segments
Customer Orientation:
- Women
- Men
- Senior Citizen
- Toddlers/Kids
Primary Ingredients:
- Vitamins
- Minerals
- Omega 3
- Botanicals Herbs or Compounds
- Prebiotics & Probiotics
- Collagen
- Amino Acid
- Enzymes
- Protein (Whey, Casein)
Form:
- Tablets & Capsules
- Gummies & Chews
- Powder
- Soft Gel
- Liquid
- Others (Lollipops, hard boiled candies)
Region:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- National Institutes of Health, Office of Dietary Supplements. (2024, July 17). Multivitamin/mineral supplements fact sheet for health professionals. U.S. Department of Health and Human Services.
- Council for Responsible Nutrition. (2024, October 29). CRN survey shows consistent supplement usage with increase of specialty product use over time.
- USA Food and Drug Administration. (2026, March 3). What’s new in dietary supplements.
- USANA Health Sciences, Inc. (2026, February 17). USANA Health Sciences reports fourth quarter and full year 2025 results and provides fiscal year 2026 outlook.
- Jamieson Wellness Inc. (2025, February 27). Jamieson Wellness Inc. reports fourth quarter and full year 2024 results.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Market intelligence to support strategic decision making across Vitamins, Minerals, Omega 3, and specialized Probiotics categories
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by raw material extraction volume tracking
- Growth opportunity mapping across specific Women and Senior Citizen demographic sub-segments with emphasis on matching continuous biomarker data with personalized dosing
- Segment and regional revenue forecasts covering Tablets & Capsules and Gummies across the localized formulation partnerships and digital-first supply chains detailed in this article
- Competition strategy assessment including supply chain transparency verification, bioavailability capabilities, and contract manufacturing capacity
- Formulation development tracking including high-speed encapsulation, repulpable blister packaging frameworks, and verifiable digital ledgers
- Market access analysis covering European Food Safety Authority directives, structured-function claim protocols, and specialized import tariff frameworks
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive strategy, retail category planning, and operational benchmarking use
Frequently Asked Questions
How big is the dietary supplements market?
The sector crosses USD 217.2 billion in 2026, signaling a profound shift away from discretionary retail purchasing toward structurally embedded, preventative wellness regimens prioritized globally by aging populations.
What is the forecast CAGR for dietary supplements through 2036?
Sustained investment pushes the valuation to USD 478.7 billion by 2036 at an 8.2% CAGR, reflecting the mass transition of specialized metabolic support formulations from niche premium channels into standard, subscription-based recurring revenue models.
Which segment leads the dietary supplements market today?
Tablets & Capsules lead entirely due to their operational superiority in shielding moisture-sensitive active ingredients while delivering the maximum allowable pharmaceutical-grade payload per serving without degrading over long logistical cycles.
Why is the dietary supplements market growing globally?
The rapid integration of continuous biomarker tracking directly links specific physiological deficits to tailored supplementation, collapsing the abandonment rate and transforming sporadic buyers into reliable subscription cohorts.
Which companies are the key players in dietary supplements?
Top-tier participants like Nestlé Health Science and Pharmavite maintain dominant positions by controlling immense formulation libraries, absorbing high capital expenditures for proprietary delivery systems, and securing priority access to high-grade raw materials.
Which countries are expanding fastest in dietary supplements?
India's 10.5% advance vastly outpaces Germany's 5.6% because the Indian trajectory is propelled by the sudden, massive deployment of digital-first direct-to-consumer networks that entirely bypass historical physical pharmacy gatekeepers.
How is e-commerce changing supplement sales and brand strategy?
Linking wearable technology or blood markers to direct fulfillment eliminates the guesswork of supplementation, creating a closed-loop digital system where specific metabolic regulators are automatically optimized and shipped based on real-time deficit data.
What role do gummies, powders, and personalized nutrition play in growth?
The shift to chewables directly solves the format fatigue inherent in multi-pill daily regimens, trading slightly lower active payloads for massively improved long-term compliance rates and personalized modular blending.
How do dietary supplement regulations by country vary across major markets?
The USA reliance on structure-function claims accelerates new product deployment without pre-market clinical mandates, whereas strict European Food Safety Authority directives force manufacturers to validate every structural claim through rigorous clinical dossiers before shelf placement.
What are the main risks for new entrants in dietary supplements?
Brands attempting to scale rapidly often hit a bottleneck when standard contract encapsulators lack the specialized equipment required to produce complex, high-bioavailability suspensions without compromising batch consistency, leading to rapid commoditization.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Customer Orientation
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Customer Orientation , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Customer Orientation , 2026 to 2036
- Women
- Men
- Pthers
- Women
- Y to o to Y Growth Trend Analysis By Customer Orientation , 2021 to 2025
- Absolute $ Opportunity Analysis By Customer Orientation , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Primary Ingredients
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Primary Ingredients, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Primary Ingredients, 2026 to 2036
- Vitamins
- Minerals
- Omega 3
- Vitamins
- Y to o to Y Growth Trend Analysis By Primary Ingredients, 2021 to 2025
- Absolute $ Opportunity Analysis By Primary Ingredients, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Tablets & Capsules
- Gummies & Chews
- Others
- Tablets & Capsules
- Y to o to Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Customer Orientation
- By Primary Ingredients
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Customer Orientation
- By Primary Ingredients
- By Form
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Customer Orientation
- By Primary Ingredients
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Customer Orientation
- By Primary Ingredients
- By Form
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Customer Orientation
- By Primary Ingredients
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Customer Orientation
- By Primary Ingredients
- By Form
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Customer Orientation
- By Primary Ingredients
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Customer Orientation
- By Primary Ingredients
- By Form
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Customer Orientation
- By Primary Ingredients
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Customer Orientation
- By Primary Ingredients
- By Form
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Customer Orientation
- By Primary Ingredients
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Customer Orientation
- By Primary Ingredients
- By Form
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Customer Orientation
- By Primary Ingredients
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Customer Orientation
- By Primary Ingredients
- By Form
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Customer Orientation
- By Primary Ingredients
- By Form
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Customer Orientation
- By Primary Ingredients
- By Form
- Competition Analysis
- Competition Deep Dive
- Bayer AG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Nestlé Health Science
- Amway
- Herbalife
- NOW Foods
- Pharmavite
- Bayer AG
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Customer Orientation , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Primary Ingredients, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Customer Orientation , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Primary Ingredients, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Customer Orientation , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Primary Ingredients, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Customer Orientation , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Primary Ingredients, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Customer Orientation , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Primary Ingredients, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Customer Orientation , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Primary Ingredients, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Customer Orientation , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Primary Ingredients, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Customer Orientation , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Primary Ingredients, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Form, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Customer Orientation , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Customer Orientation , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Customer Orientation
- Figure 6: Global Market Value Share and BPS Analysis by Primary Ingredients, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Primary Ingredients, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Primary Ingredients
- Figure 9: Global Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Form
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Customer Orientation , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Customer Orientation , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Customer Orientation
- Figure 26: North America Market Value Share and BPS Analysis by Primary Ingredients, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Primary Ingredients, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Primary Ingredients
- Figure 29: North America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Form
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Customer Orientation , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Customer Orientation , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Customer Orientation
- Figure 36: Latin America Market Value Share and BPS Analysis by Primary Ingredients, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Primary Ingredients, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Primary Ingredients
- Figure 39: Latin America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Form
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Customer Orientation , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Customer Orientation , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Customer Orientation
- Figure 46: Western Europe Market Value Share and BPS Analysis by Primary Ingredients, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Primary Ingredients, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Primary Ingredients
- Figure 49: Western Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Form
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Customer Orientation , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Customer Orientation , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Customer Orientation
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Primary Ingredients, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Primary Ingredients, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Primary Ingredients
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Form
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Customer Orientation , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Customer Orientation , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Customer Orientation
- Figure 66: East Asia Market Value Share and BPS Analysis by Primary Ingredients, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Primary Ingredients, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Primary Ingredients
- Figure 69: East Asia Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Form
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Customer Orientation , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Customer Orientation , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Customer Orientation
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Primary Ingredients, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Primary Ingredients, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Primary Ingredients
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Customer Orientation , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Customer Orientation , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Customer Orientation
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Primary Ingredients, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Primary Ingredients, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Primary Ingredients
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

