The global cell therapy manufacturing market is estimated to be valued at USD 6.2 billion in 2026 and is projected to reach USD 23.2 billion by 2036, growing at a CAGR of 14.2%. Based on Future Market Insights’ estimations, cell therapy manufacturing industry is driven by a fundamental paradigm shift toward standardized, scalable commercial processes that replace fragmented academic-style production. Regulatory bodies are accelerating this trajectory; the US FDA (January 2026) introduced new reforms to expedite Chemistry, Manufacturing, and Controls (CMC) reviews. This initiative allows for greater flexibility in commercial specifications, directly addressing the bottleneck of rigid post-approval change requirements.
As per FMI's projection, regulatory easing will reduce time-to-market for autologous therapies by approximately 18-24 months. Industry stakeholders are currently transitioning from a "scientific discovery" phase into an "industrial efficiency" phase, where cost-of-goods-sold (COGS) reduction becomes the primary competitive metric.
”Regulatory flexibility must be tailored for cell and gene therapies. These are common-sense reforms that will address the unique characteristics of cell and gene therapies and foster more innovation.” - Dr. Marty Makary, Commissioner, US Food and Drug Administration (FDA). The perception points at the critical transition from rigid compliance frameworks to adaptive regulatory pathways that favor rapid commercialization.
Manufacturers face a distinct opportunity to capitalize on the "capacity gap" as patent cliffs push big pharma toward outsourced models. Despite high growth, the sector grapples with immense financial pressure; production costs for a single CAR-T dose often exceed USD 80,000, creating friction with payers demanding value-based reimbursement. Data from the Cell and Gene Therapy Catapult (2025) indicates that 45% of manufacturing deviations arise from manual handling errors, costing the industry millions annually. Companies are responding by investing in closed-system automation and digital twins to minimize human intervention.
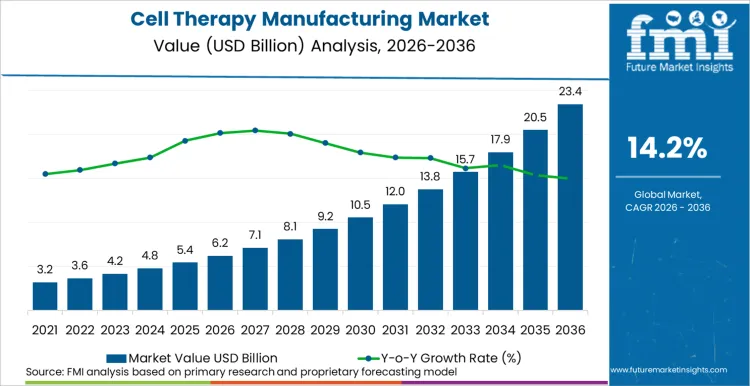
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 6.2 Billion |
| Industry Value (2036) | USD 23.2 Billion |
| CAGR (2026-2036) | 14.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Unified guidelines across major jurisdictions are significantly reducing the friction associated with post-approval manufacturing changes. During 2020 to 2025, minor process tweaks required re-submission of data in every geography, stalling global rollouts. FMI analysis suggests that new frameworks, such as the FDA's 2026 CMC reforms, will allow manufacturers to implement efficiency upgrades without triggering full regulatory holds. This shift is critical for maintaining inventory levels; companies can - adapt to supply chain shortages or upgrade to superior reagents faster. Corporate data from Kite Pharma (January 2024) validates this, as process optimizations approved by regulators successfully reduced their median turnaround time to just 14 days, directly impacting patient survival rates.
Contamination risks in open manual processing remain the single largest threat to batch viability and patient safety. Automated, closed systems are becoming the standard of care, driven by the need to eliminate "human error" variables. Statistics from industry pilots show that closed-process automation can reduce contamination rates by over 90% while simultaneously lowering labor costs. This trend is accelerating investment in bioprocessing equipment. Sartorius (February 2025) reported that demand weakness has ended, signaling a renewed capex cycle where manufacturers replace legacy open suites with integrated, automated platforms to ensure commercial robustness.
The cell therapy manufacturing market is segmented by manufacturing scale, technology used, and mode of production. By scale, manufacturing is categorized into pre clinical, clinical scale, and commercial scale based on development stage and production volume.
By technology, the market includes viral vector production, non viral vector production, cell processing equipment, and process development activities supporting cell expansion and modification.
By mode, manufacturing is conducted either in house by therapy developers or outsourced to contract manufacturing organizations CDMOs.
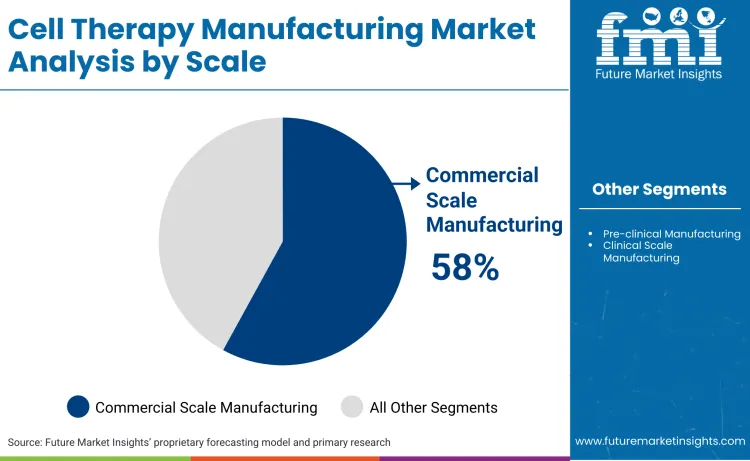
Commercial scale manufacturing commands 58% share in 2026, driven by the surging number of FDA-approved cell therapies entering routine clinical practice. Manufacturers can capitalize on the dominance of commercial scale manufacturing by prioritizing early commercial readiness rather than treating scale up as a late stage activity. Capacity must be designed for continuous operations, with modular cleanrooms, parallel processing lines, and validated redundancy built in from the outset. Competitive advantage is created when manufacturers demonstrate consistent GMP performance, low batch failure rates, and predictable release timelines, as buyers increasingly favor reliability over price. Long term supply contracts should be targeted with approved therapy sponsors, locking in utilization and stabilizing cash flows. Strategic investments in automation, closed system processing, and digital batch records reduce labor dependency and improve audit outcomes. Manufacturers that integrate downstream logistics, cryogenic storage, and last mile delivery capabilities are positioned to capture premium pricing. Entry barriers rise sharply at this stage, allowing incumbents to defend margins while new entrants struggle with regulatory credibility, scale economics, and late stage quality expectations.
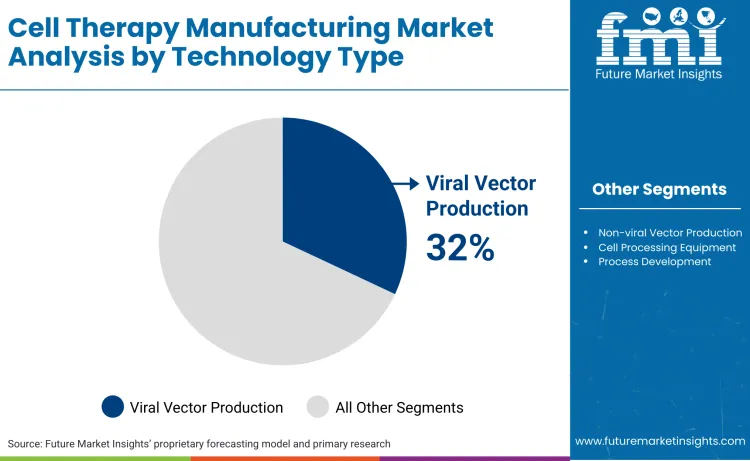
Viral vector technologies account for 32% share in 2026. Manufacturers can strengthen their position in viral vector technologies by focusing on yield improvement, failure rate reduction, and platform standardization. Demand remains supply constrained, enabling premium pricing for reliable vector output. Investment in suspension based producer cell lines, optimized plasmid ratios, and scalable bioreactor systems directly improves cost per dose and contract attractiveness. Manufacturers should position viral vector capacity as a bottleneck solution rather than a commodity service, offering guaranteed slots and performance backed timelines. Long term master service agreements with cell therapy developers reduce volatility and discourage switching. Vertical integration into plasmid DNA production or downstream purification further protects margins and mitigates supply risk. As non viral approaches remain clinically selective, viral vectors retain regulatory familiarity and approval momentum.
Regional manufacturing is being treated as a risk containment mechanism rather than a margin optimization lever. Trade uncertainty, tariff exposure, and policy volatility are increasing operational fragility for centralized production hubs. Manufacturing footprints are being redesigned to serve key consumption markets from within the same geography to reduce regulatory exposure and supply disruption risk.
Dr Reddys Laboratories illustrated this approach in May 2025 through evaluation of United States manufacturing sites while retaining Indian operations. The structure supports continuity in regulated markets while preserving cost anchored production. Local for local manufacturing is influencing site selection logic, compliance planning, and long term capital deployment across pharmaceutical supply chains.
End to end integration is being adopted to remove coordination inefficiencies across discovery, development, manufacturing, and commercialization. Fragmented operating structures often introduce delays through external handoffs, misaligned accountability, and quality variability. Integrated models consolidate control under a single governance framework, enabling faster execution and tighter process discipline.
Kite Pharma demonstrated this model in 2024 by operating a fully integrated network that delivered industry leading turnaround times in cell therapy production. Control over critical steps enabled faster patient delivery while maintaining compliance thresholds. Integrated execution is being positioned as a competitive differentiator in complex biologics.
Outsourcing is being positioned as a capital risk management tool rather than a structural replacement for internal manufacturing. Large pharmaceutical sponsors are delaying fixed asset investment until late stage pipeline validation improves certainty. External capacity allows scaling without long term balance sheet exposure or asset underutilization risk.
Fujifilm Diosynth stated in January 2025 that restrained internal investment by large sponsors is accelerating demand for contract manufacturing expansion. Outsourcing is functioning as a volatility absorber during pipeline evaluation cycles while supporting flexibility in capacity planning.
China at 12.1% shows the highest expansion rate, supported by domestic capacity expansion and localized manufacturing programs. India at 11.0% follows, driven by cost efficient production models and increasing participation in outsourced manufacturing. The United States at 10.0% reflects steady progress supported by an established CDMO ecosystem and late stage therapy activity. The United Kingdom at 9.5% maintains momentum through focused cell and gene therapy clusters. Germany at 7.9% exhibits measured growth influenced by quality centric production and regulatory rigor. Japan at 6.0% records the lowest CAGR due to longer commercialization timelines and cautious adoption patterns. China records the highest growth among the listed countries, while Japan records the lowest relative to the global benchmark.
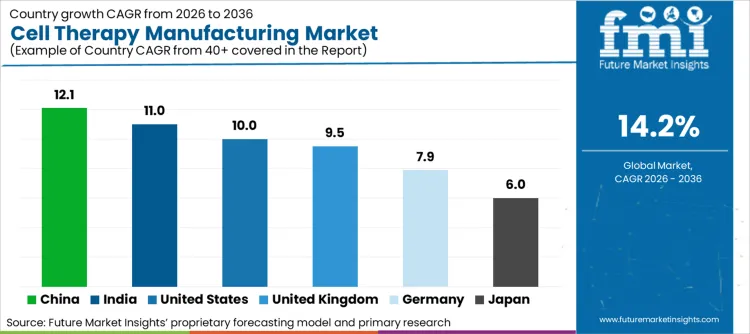
| Country | CAGR (2026-2036) |
|---|---|
| India | 11.0% |
| Germany | 7.9% |
| United States | 10.0% |
| United Kingdom | 9.5% |
| China | 12.1% |
| Japan | 6.0% |
The cell therapy manufacturing market in the United States are set to rise at a solid 10.0% CAGR. Growth is underpinned by a regulatory pivot toward manufacturing flexibility intended to maintain American leadership in biotechnology. The FDA (January 2026) announced a new flexible approach to CMC requirements to support innovation. Kite Pharma exemplified this progress by securing approval for a process that reduces turnaround time to 14 days, setting a new commercial standard. Dr. Marty Makary, Commissioner of the FDA, validated this shift: 'Regulatory flexibility must be tailored for cell and gene therapies. These are common-sense reforms that will address the unique characteristics of cell and gene therapies and foster more innovation.’ This strategic pivot signals a move away from rigid compliance frameworks that previously stifled speed.
Demand for cell therapy manufacturing in China is anticipated to grow at 12.1% CAGR. Rapid expansion is supported by standardized scale-up processes and massive infrastructure investment. The NMPA (2025) released updated technical guiding principles for pharmaceutical change research, establishing clearer classification systems for manufacturing changes. WuXi AppTec reported in October 2025 that it expects to resume double-digit revenue growth, signaling a recovery in the region's massive CDMO infrastructure. The Management Team at WuXi AppTec noted: 'The Company expects Continuing Operations revenue to resume double-digit growth in 2025, with its year-over-year growth rate raised to 17-18%, up from the prior 13-17%.' Such confidence reflects a broader industrial resilience that defies external market headwinds.
The cell therapy manufacturing industry in the United Kingdom is projected to expand at 9.5% CAGR. This robust growth is underpinned by a strong clinical pipeline and government support. The Cell and Gene Therapy Catapult (2025) reported continued growth in early-stage trials, directly feeding demand for local manufacturing slots. Oxford Biomedica (April 2025) confirmed its pivot to profitability, validating the region's maturity from an R&D hub to a commercial manufacturing center. Dr. Frank Mathias, CEO of Oxford Biomedica, stated: 'Through continued focus on efficiency and a disciplined approach to our cost base, we also expect to achieve operating EBITDA profitability for FY 2025.' This milestone marks a decisive transition from subsidized research toward financially sustainable industrial operations. The UK is thus cementing its status as the premier European destination for complex viral vector supply chains.
Cell therapy manufacturing market in Germany is poised to register a 7.9% CAGR. Germany's strength lies in its engineering heritage, making it the central hub for bioprocessing equipment and supply chain stability. Rapid expansion of advanced therapy manufacturing infrastructure is reshaping global capacity dynamics, with new large‑scale facilities signalling a shift toward more robust, standardized production ecosystems. A notable example is the recent opening of ProBio’s flagship plasmid and viral vector manufacturing center in New Jersey, which underscores the sector’s accelerating investment in high‑quality, scalable bioprocessing capabilities. As Allen Guo, CEO of ProBio, emphasized during the facility’s launch, “Opening the Hopewell center is a milestone in ProBio's global expansion and underscores our mission to support biopharma innovators with high-quality, responsive manufacturing solutions.” This sentiment shows a broader industry commitment to bridging the gap between scientific breakthroughs and commercial‑ready manufacturing, ensuring that next‑generation cell and gene therapies can move more swiftly and reliably from development to patients worldwide.
Sales of Cell Therapy Manufacturing in India are forecast to grow at 11.0% CAGR. India is leveraging its renewable energy capacity and cost advantages to attract global partnerships. The Ministry of New and Renewable Energy (November 2025) reported record capacity additions, supporting energy-intensive biomanufacturing. Dr. Reddy's Laboratories (May 2025) emphasized its strategy to leverage Indian manufacturing for global markets while remaining open to US production to mitigate tariff risks. Erez Israeli, CEO of Dr. Reddy's, stated: 'We are more than open to work in the United States, to make in the United States when the opportunity comes.'
Demand for cell therapy manufacturing in Japan is set to rise at 6.0% CAGR. Stability is driven by long-term government promotion of regenerative medicine and CDMO investment. The PMDA (December 2024) was designated as a research institution to promote "Regulatory Science," accelerating practical applications. Fujifilm Diosynth Biotechnologies continues to expand its global ecosystem from its Japanese base, as confirmed in its January 2025 update on its USD 8 billion investment plan. Lars Petersen, President & CEO of Fujifilm Diosynth, observed: 'I still think, like many others, that this is the future. My take is that a lot of the large pharma companies, they didn't want to invest themselves, so they prefer to outsource more.' This commitment to heavy capital investment signals a long-term belief in the outsourcing model's durability. Japan is subsequently positioning itself as the high-quality, investment-heavy anchor for the Asian advanced therapy market.
Leading CDMOs are aggressively expanding capacity to capture the outsourcing wave from big pharma. FMI analysis indicates that the leading players such as Lonza, Thermo Fisher, WuXi AppTec, Catalent, and Fujifilm, control significant market share through their "end-to-end" service offerings. Their dominance is based on the ability to handle everything from plasmid design to commercial fill-finish. Consolidation is a key trend; Fujifilm's USD 8 billion investment plan (January 2025) exemplifies the massive capital requirements now needed to compete. Smaller players are being forced to specialize in niche technologies like viral vectors or specific cell types to survive against these integrated giants.
Companies are pivoting from pure capacity expansion to efficiency and profitability. Oxford Biomedica's shift to EBITDA profitability in 2025 illustrates this strategic discipline. Manufacturers are also pursuing geographic diversification to hedge against trade barriers. Dr. Reddy's willingness to establish US manufacturing in response to tariff threats demonstrates this agility. Cindy Perettie, EVP at Kite Pharma, emphasized the importance of integrated speed: 'We're the only company that has end-to-end standalone research and development, manufacturing and commercialisation of a potentially curative treatment.' This response highlights that vertical integration is no longer just a luxury but a defensive moat.
The cell therapy manufacturing market represents revenue generated from the production of cell-based therapeutic products and associated manufacturing services, as operationally defined in the article. The market measures commercial and clinical manufacturing activities required to produce cell therapies under regulated conditions, including process development, scale-up, and GMP-compliant production. Market sizing reflects manufacturing-stage revenues captured by in-house facilities and outsourced contract development and manufacturing organizations (CDMOs), analysed by manufacturing scale, technology type, mode of production, and region, and expressed in USD billion.
The market includes cell therapy manufacturing conducted at pre-clinical, clinical, and commercial scales, with commercial-scale manufacturing identified as the dominant revenue segment. It covers manufacturing technologies explicitly analysed in the article, including viral vector production, non-viral vector technologies, and cell processing activities. Revenue from both in-house manufacturing and outsourced CDMO services is included. The scope counts manufacturing activities supporting autologous and allogeneic cell therapies, including CAR-T and other advanced therapies, where value is generated through capacity utilization, process robustness, automation, and scalability. Manufacturing investments in closed-system processing, automation, digital twins, and end-to-end production platforms are included when they directly support cell therapy manufacturing output. The market covers revenues generated across North America, Europe, East Asia, South Asia, Latin America, and the Middle East & Africa.
The market excludes revenues from cell therapy discovery research, preclinical biological research not tied to manufacturing output, and purely academic or laboratory experimentation. Clinical trial outcomes, therapeutic efficacy revenues, and drug sales to patients or healthcare systems are not included. Upstream raw material production, reagent sales, and bioprocessing equipment sales are excluded unless captured as part of manufacturing service revenue. Regulatory approvals, policy initiatives, and public funding referenced in the article are not counted as market revenue. Logistics, distribution, commercialization, and post-marketing pharmacovigilance activities are also outside the scope of the cell therapy manufacturing market.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 6.2 billion |
| Manufacturing Scale | Pre-clinical Manufacturing, Clinical Scale Manufacturing, Commercial Scale Manufacturing |
| Technology Type | Viral Vector Production, Non-viral Vector Production, Cell Processing Equipment, Process Development |
| Mode of Production | In-house Manufacturing, Contract Manufacturing (CDMO) |
| Therapy Type | Autologous Cell Therapies, Allogeneic Cell Therapies, CAR-T Therapies, Advanced Therapy Medicinal Products (ATMPs) |
| End-Use In dustry | Biopharmaceutical Companies, Academic & Research Institutions, Hospitals & Treatment Centers, Contract Development and Manufacturing Organizations |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | United States, United Kingdom, Germany, India, Japan, China, and 30+ countries |
| Key Companies Profiled | Lonza Group, Thermo Fisher Scientific, WuXi AppTec, Miltenyi Biotec, Sartorius AG, Boehringer Ingelheim, Fujifilm Diosynth Biotechnologies, Kite Pharma (Gilead Sciences), Novartis AG, Roche, Charles River Laboratories, Oxford Biomedica |
| Additional Attributes | Dollar sales by manufacturing scale and technology type, regional demand trends, competitive landscape analysis, CDMO outsourcing strategies, closed-system automation adoption, GMP compliance frameworks, cost-of-goods-sold (COGS) optimization, regulatory pathway evolution, capacity expansion investments, and end-to-end integrated manufacturing models |
Source: FMI historical analysis and forecast data.
What is the current global market size for Cell Therapy Manufacturing?
Global market is valued at USD 6.2 billion in 2026, driven by the commercialization of CAR-T therapies.
What is the projected Compound Annual Growth Rate (CAGR) for the market over the next 12 years?
According to FMI's estimates, the market is projected to grow at 14.2% CAGR from 2024 to 2036.
Which technology segment is the primary driver of demand?
Viral vector production holds the leading share of 32% due to high complexity and outsourcing rates.
What are the primary market risks or regulatory hurdles?
High cost of goods sold (COGS) and stringent CMC requirements create significant barriers to scalable profitability.
Who are the leading suppliers in the CDMO space?
Lonza, WuXi AppTec, and Fujifilm Diosynth lead the market through extensive global capacity and integrated service models.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.