Carpal Tunnel Release System Market
The Carpal Tunnel Release System Market is Segmented by Product Types (Open CTR System, Endoscopic CTR System) and End User (Hospitals, Ambulatory Surgical Centers, Specialty Clinics). Forecast for 2026 to 2036.
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Carpal Tunnel Release System Market Size, Market Forecast and Outlook By FMI
The carpal tunnel release system market is expected to expand from USD 702.0 million in 2026 to USD 1,199.1 million by 2036. The market is anticipated to register a 5.5% CAGR during the forecast period. Open CTR System is likely to lead product types with a 62.0% share, while hospitals are projected to account for 60.0% share of the end user segment in 2026.
Summary of the Carpal Tunnel Release System Market
- Demand and Growth Drivers
- Rising carpal tunnel syndrome cases are expected to support procedure volumes across hospital and outpatient surgical settings.
- Wider awareness of wrist nerve compression symptoms is likely to improve treatment referrals and surgical evaluation rates.
- Minimally invasive procedure interest is anticipated to increase device use in settings focused on faster patient recovery.
- Product and Segment View
- Open CTR System is expected to lead the product types segment due to surgeon familiarity and established procedure use.
- Hospitals are likely to remain the main end user because complex cases need specialized teams and broader surgical infrastructure.
- Endoscopic CTR systems are anticipated to gain attention as providers seek smaller-incision options for suitable patients.
- Geography and Competitive Outlook
- India is expected to lead country-level expansion due to improving healthcare access and rising treatment awareness.
- China is likely to record steady expansion as urban healthcare systems increase access to surgical procedures.
- Companies with stronger surgical device portfolios and clinician training support are anticipated to improve competitive positioning.
- Analyst Opinion
- Sabyasachi Ghosh, Principal consultant for Healthcare at FMI, suggests, “The carpal tunnel release system market is likely to move toward procedure-ready systems that fit both hospital and ambulatory workflows. Established surgical device companies are expected to retain an advantage as clinicians prioritize reliability, training support, and predictable patient outcomes.”
- Carpal Tunnel Release System Market Value Analysis
- The carpal tunnel release system market is moving from conventional open surgery reliance toward a broader device mix.
- Procedure demand is likely to rise as carpal tunnel syndrome recognition improves across working and aging populations.
- Use is expected to increase in hospitals where surgical teams manage higher case volumes and complex patient needs.
- Spending is anticipated to be supported by outpatient procedure expansion and investment in minimally invasive surgical tools.
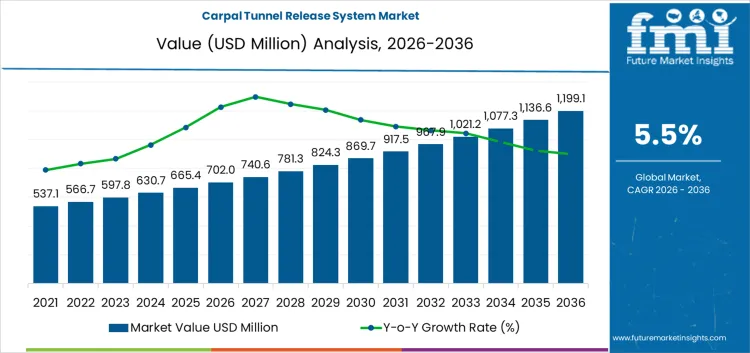
What is the Growth Forecast for the Carpal Tunnel Release System Market through 2036?
The acceleration and deceleration pattern of the carpal tunnel release system market reveals an initial phase of steady acceleration followed by a more gradual growth rate as the market matures. From 2026 to 2031, the market will grow from USD 702 million to USD 967.9 million, reflecting a steady increase of USD 265.9 million. During this phase, there will be an acceleration in market growth due to the increasing prevalence of CTS, growing adoption of minimally invasive treatment options, and technological advancements that improve the effectiveness and accessibility of carpal tunnel release procedures. The early growth will be driven by heightened awareness of CTS, as well as the increasing number of patients seeking treatment.
From 2031 to 2036, the market will continue to expand, increasing from USD 967.9 million to USD 1,199.1 million, reflecting an additional USD 231.2 million in value. However, the rate of growth will decelerate in the later years of the forecast period. This deceleration is typical of maturing markets, where the treatment options become more widely available, and market growth stabilizes. During this phase, the market will shift from rapid adoption of new treatments to optimizing existing ones and expanding access to care. While the market will continue to grow, it will do so at a more stable pace, driven by incremental improvements in treatment technologies and expanding healthcare access.
Carpal Tunnel Release System Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Sales Value (2026) | USD 702 million |
| Industry Forecast Value (2036) | USD 1,199.1 million |
| Industry Forecast CAGR (2026 to 2036) | 5.5% |
What Is Driving the Demand for the Carpal Tunnel Release System Market?
Historical demand for carpal tunnel release systems has been shaped by the increasing prevalence of carpal tunnel syndrome (CTS), a condition caused by compression of the median nerve in the wrist that leads to pain, numbness, and impaired hand function. CTS became more recognized as work related musculoskeletal disorder prevalence rose among working populations, driven by repetitive hand movements, prolonged computer use, and ageing demographics, prompting healthcare providers to seek effective surgical interventions.
Traditional open carpal tunnel release techniques were widely adopted because they reliably relieved nerve pressure, supporting growth in demand for devices and instruments used in these procedures. Over time, awareness of minimally invasive alternatives such as endoscopic carpal tunnel release systems grew among surgeons and patients because these options offered smaller incisions and faster recovery with reduced postoperative discomfort. The historical market benefited from rising healthcare expenditure, expansion of surgical facilities, and development of specialized surgical tools that enhanced treatment outcomes and broadened access for patients in clinical settings.
Future demand for carpal tunnel release systems is expected to be driven by continued increases in CTS prevalence, expansion of minimally invasive surgical adoption, and technological innovation in surgical instruments. Market forecasts suggest growth from around USD 702 million in 2025 to approximately USD 1,199.1 million by 2035 at about 5.5 % CAGR, reflecting rising demand for advanced systems that support efficient procedures and improved patient recovery.
Surgeons and healthcare facilities are increasingly adopting endoscopic devices, ultrasound guided systems, and instrumentation that support outpatient and ambulatory surgical center workflows, enhancing procedure throughput and patient experience. An ageing global population, expanding awareness of CTS symptoms and treatment options, and broader healthcare access in emerging markets also contribute to future growth. Manufacturers are investing in research and development to deliver next generation devices with improved ergonomics, imaging integration, and automation capabilities that further drive market uptake and provide clinicians with tools suited to evolving procedural standards.
What are the Leading Segments in the Global Carpal Tunnel Release System Market?
The global carpal tunnel release system market is segmented by product type and end user. Among product types, open CTR system leads the market, capturing 62% of the share, followed by endoscopic CTR system. Open CTR systems are preferred for their direct approach to treating carpal tunnel syndrome, offering surgeons a more straightforward method for the release procedure. On the end-user side, hospitals dominate the market, holding 60% of the share, followed by ambulatory surgical centers and specialty clinics. Hospitals lead due to the wide availability of specialized equipment and expertise in treating carpal tunnel syndrome, as well as the greater volume of surgeries performed in hospital settings.
What Drives the Growth of Open CTR Systems in the Carpal Tunnel Release System Market?
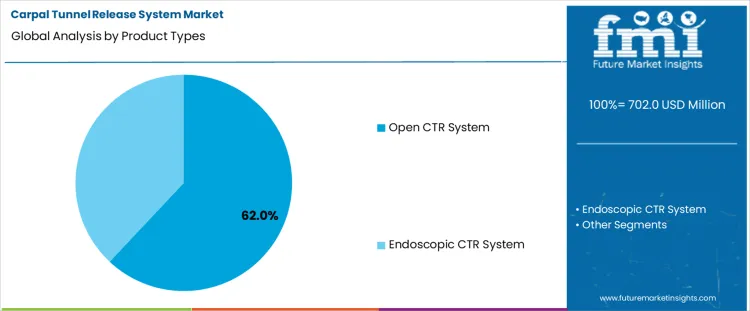
Open CTR systems are the leading product type in the carpal tunnel release system market, holding 62% of the share. This demand is driven by the direct and widely used surgical approach they offer for carpal tunnel release. Open CTR systems involve a single incision to access the carpal tunnel, which allows for greater visibility and direct release of the median nerve. This method has been traditionally preferred due to its established effectiveness and the ease with which surgeons can perform the procedure, particularly for more severe cases of carpal tunnel syndrome. While endoscopic techniques are gaining popularity for their minimally invasive nature, the open CTR system remains dominant in the market due to its well-documented success and surgeon familiarity. Additionally, it is often more accessible in hospitals with broader surgical capabilities. As carpal tunnel syndrome remains a common condition requiring surgical intervention, the continued preference for open CTR systems, especially in complex or complicated cases, will sustain their market leadership.
What is Driving the Demand for Hospitals in the Carpal Tunnel Release System Market?
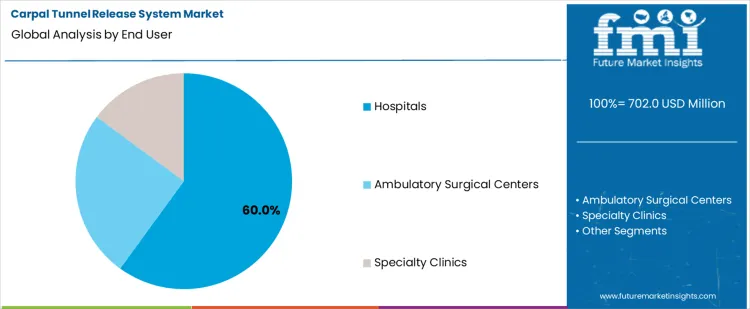
Hospitals are the leading end-user in the carpal tunnel release system market, capturing 60% of the share. This growth is primarily driven by the high volume of surgeries performed in hospitals, where specialized surgical teams and advanced equipment are readily available. Hospitals are equipped with the necessary infrastructure to handle both inpatient and outpatient carpal tunnel release procedures, making them the preferred setting for more complex cases. The presence of skilled surgeons and a comprehensive range of post-operative care options further boosts the demand for carpal tunnel release systems in hospital environments. Hospitals also have the resources to support the latest surgical techniques, including both open and endoscopic CTR systems, and can offer a full continuum of care, from diagnosis to rehabilitation. As the incidence of carpal tunnel syndrome continues to rise, hospitals will remain a dominant force in the market, providing the expertise and resources needed for successful treatment.
What are the Key Dynamics in the Carpal Tunnel Release System Market?
The carpal tunnel release system market is shaped by demand for surgical technologies that treat median nerve compression in the wrist with precision and safety. These systems include open, endoscopic, and minimally invasive devices designed to facilitate controlled release of the transverse carpal ligament. Adoption depends on clinical evidence, surgeon preference, system compatibility with hospital infrastructure, and patient outcomes. Regulatory frameworks for medical devices influence approval and uptake. Cost structures, reimbursement policies, and training requirements for healthcare professionals affect how systems are integrated into orthopaedic, neurosurgical, and outpatient surgical settings. Competition among established and emerging suppliers influences pricing and innovation.
What are the Main Drivers Supporting Growth in This Market?
Growth in the carpal tunnel release system market is driven by increasing prevalence of carpal tunnel syndrome linked to aging populations and repetitive motion activities. Rising awareness of early diagnosis and treatment options encourages specialist referrals and surgical intervention. Advances in minimally invasive and endoscopic technologies support shorter recovery times and reduced postoperative discomfort, which appeal to clinicians and patients alike. Expansion of outpatient surgical facilities and ambulatory care centres enhances access to procedures. Training programmes and clinical evidence demonstrating efficacy and safety of new systems support surgeon confidence. Health systems focused on improving functional outcomes and reducing complication rates are increasingly adopting advanced release systems.
What Restraints Are Limiting Market Expansion?
The market faces restraints related to cost, training, and healthcare infrastructure. High acquisition and maintenance costs of advanced carpal tunnel release systems can deter smaller hospitals and clinics, particularly in cost sensitive regions. Endoscopic and minimally invasive systems require specific training and a learning curve for surgeons, which may slow adoption where educational resources are limited. Variability in reimbursement policies for surgical devices and procedures across regions influences purchase decisions. Some clinicians continue to rely on conventional open release techniques due to familiarity and established outcomes, reducing demand for alternative systems. Concerns about procedural complications in inexperienced hands can also restrain broader uptake.
What Emerging Trends Are Shaping the Future of This Market?
Emerging trends include continued refinement of endoscopic and ultrasound guided release techniques that aim to enhance precision and reduce tissue trauma. Integration of digital planning tools and intraoperative imaging improves visualization and procedural confidence. Manufacturers are focusing on ergonomics and single use components to streamline workflows and reduce infection risk. There is growing interest in bundled care pathways that link device choice with rehabilitation and functional recovery metrics. Training models using simulation and virtual reality are gaining traction to shorten the learning curve for new technologies. Value based healthcare initiatives that emphasise patient reported outcomes and cost effectiveness are increasingly influencing adoption of advanced carpal tunnel release systems.
What is the Country-Wise Analysis of the Carpal Tunnel Release System Market?
The carpal tunnel release system market is experiencing steady growth due to increasing awareness of carpal tunnel syndrome (CTS) and advancements in surgical techniques. Developed markets such as Germany, France, and South Korea are witnessing moderate growth, with rising demand for minimally invasive treatments and better surgical outcomes.
Emerging markets like India and China are also showing steady growth, driven by a large population base and increasing healthcare access. Factors such as aging populations, the rise in repetitive strain injuries due to sedentary lifestyles, and technological advancements in medical devices are driving the market. As awareness increases and healthcare infrastructure improves, the carpal tunnel release system market is expected to grow globally.
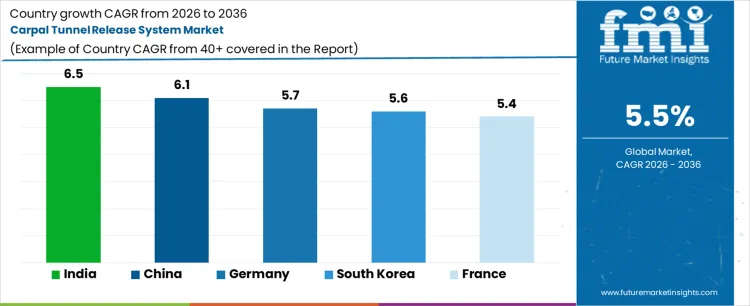
| Country | CAGR 2026 to 2036 |
|---|---|
| India | 6.5% |
| China | 6.1% |
| South Korea | 5.6% |
| Germany | 5.7% |
| France | 5.4% |
What is the Growth Outlook for the Carpal Tunnel Release System Market in India?
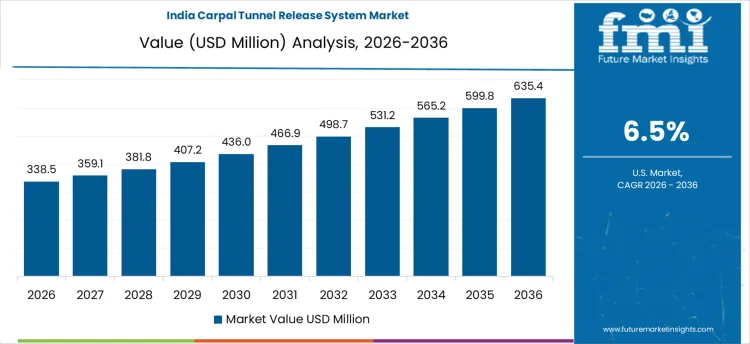
The carpal tunnel release system market in India is projected to grow steadily, with a projected CAGR of 6.5% from 2026 to 2036. India, with its vast population and increasing prevalence of carpal tunnel syndrome (CTS), is witnessing a rise in the demand for carpal tunnel release systems. This is largely due to factors such as increasing healthcare access, rising awareness of CTS, and a growing aging population. Additionally, India’s healthcare sector is expanding rapidly, leading to greater availability of advanced surgical treatments for CTS. The rise in lifestyle-related conditions, including repetitive strain injuries from technology use and sedentary jobs, is also contributing to the demand for carpal tunnel release systems. As medical technology continues to improve and healthcare infrastructure expands, the market for carpal tunnel release systems in India is expected to grow significantly, particularly in urban areas with access to advanced medical facilities.
What is the Market Outlook for the Carpal Tunnel Release System Market in China?
China is expected to experience steady growth in the carpal tunnel release system market, with a projected CAGR of 6.1% from 2026 to 2036. China’s large population and growing awareness of carpal tunnel syndrome (CTS) are key factors driving the demand for carpal tunnel release systems. As the country’s healthcare infrastructure improves, more people are seeking medical treatment for CTS, especially in urban areas where healthcare access is becoming more widely available. The rise in cases of repetitive strain injuries, particularly due to the increasing use of technology and sedentary lifestyles, is further contributing to the market’s growth. In addition, China is witnessing an increasing adoption of minimally invasive surgical procedures, which is boosting the demand for carpal tunnel release systems. As the Chinese government continues to focus on improving public healthcare and access to medical treatments, the carpal tunnel release system market is expected to see sustained growth in the coming years.
What is the Growth Forecast for the Carpal Tunnel Release System Market in South Korea?
South Korea is projected to experience moderate growth in the carpal tunnel release system market, with a projected CAGR of 5.6% from 2026 to 2036. South Korea has a well-established healthcare system, and the demand for carpal tunnel release systems is growing due to an increasing prevalence of carpal tunnel syndrome (CTS), especially among the working population. The rise in sedentary work practices, combined with the widespread use of electronic devices, is contributing to the increase in CTS cases. South Korean consumers are becoming more aware of CTS and the surgical options available, leading to a higher demand for minimally invasive procedures such as carpal tunnel release surgery. As the healthcare sector continues to develop and improve, the availability of advanced treatment options for CTS will likely drive the growth of the carpal tunnel release system market in South Korea.
What is the Market Outlook for the Carpal Tunnel Release System Market in Germany?
Germany is expected to experience steady growth in the carpal tunnel release system market, with a projected CAGR of 5.7% from 2026 to 2036. As one of Europe’s leading healthcare markets, Germany’s demand for carpal tunnel release systems is increasing due to the rising prevalence of carpal tunnel syndrome (CTS), particularly in the aging population and among workers in professions that require repetitive hand movements. Germany’s advanced healthcare infrastructure and high levels of medical awareness are contributing to the growing demand for carpal tunnel release systems. Additionally, the increasing adoption of minimally invasive surgical techniques is boosting the use of carpal tunnel release systems. As healthcare technology continues to evolve and more people seek treatments for CTS, the carpal tunnel release system market in Germany is expected to grow steadily, supported by continued advancements in medical devices and surgical techniques.
What is the Growth Forecast for the Carpal Tunnel Release System Market in France?
The carpal tunnel release system market in France is expected to grow at a steady pace, with a projected CAGR of 5.4% from 2026 to 2036. France has a well-established healthcare system and a growing awareness of carpal tunnel syndrome (CTS) among the population. The increasing prevalence of CTS, particularly among older adults and individuals with high-risk occupations such as those in manufacturing or office-based work, is driving demand for carpal tunnel release systems. France’s focus on healthcare accessibility and the rise of minimally invasive treatments are key factors contributing to the growth of the market. Additionally, the growing number of surgical procedures performed for CTS is likely to lead to an increase in the adoption of carpal tunnel release systems. As the healthcare industry continues to innovate and improve, the market for carpal tunnel release systems in France is expected to grow steadily in the coming years.
What is the Competitive Landscape for the Carpal Tunnel Release System Market?
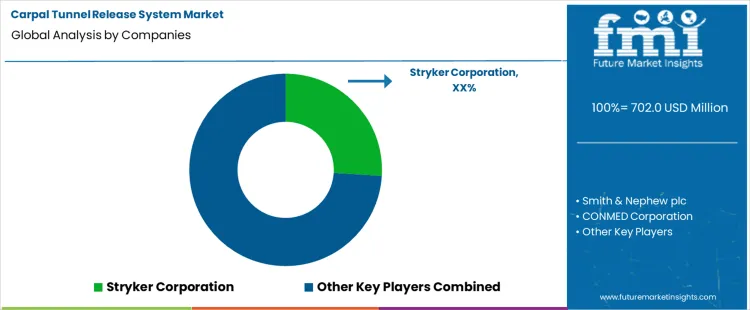
The carpal tunnel release system market is growing as surgical techniques and medical technologies advance, offering more effective and minimally invasive solutions for patients suffering from carpal tunnel syndrome. Stryker Corporation leads the market with its innovative carpal tunnel release systems, providing high-quality, precision surgical instruments that enhance surgical outcomes and recovery times.
Their focus on cutting-edge technology, product reliability, and surgical efficacy has positioned them as a dominant player in the market. Smith & Nephew plc, CONMED Corporation, and Arthrex Inc. are significant competitors, each offering specialized carpal tunnel release systems with advanced features. Smith & Nephew focuses on developing instruments that support minimally invasive surgeries, while CONMED and Arthrex Inc. emphasize high-performance tools for carpal tunnel release procedures.
Integra LifeSciences, MicroAire Surgical Instruments LLC, and S.E.G-WAY Orthopaedics Inc. further contribute to the competitive landscape by offering their own solutions for carpal tunnel release. Integra LifeSciences specializes in advanced surgical instruments and technologies that improve precision and patient outcomes, while MicroAire Surgical Instruments LLC focuses on innovative surgical devices for a range of orthopaedic procedures. S.E.G-WAY Orthopaedics Inc. offers specialized instruments designed for carpal tunnel release surgery.
A.M. Surgical Inc., Sonex Health LLC, and Innomed Inc. also play key roles in the market by providing innovative, minimally invasive solutions that cater to the needs of surgeons and patients. These companies compete by focusing on product innovation, surgical precision, and the ability to meet the growing demand for efficient, minimally invasive carpal tunnel release systems. As the market for carpal tunnel release systems continues to grow, competition and technological advancements are expected to drive further innovation in the sector.
Key Players of the Carpal Tunnel Release System Market
- Stryker Corporation
- Smith & Nephew plc
- CONMED Corporation
- Arthrex Inc.
- Integra LifeSciences
- MicroAire Surgical Instruments LLC
- S.E.G-WAY Orthopaedics Inc.
- A.M. Surgical Inc.
- Sonex Health LLC
- Innomed Inc.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| Product Types | Open CTR System, Endoscopic CTR System |
| End User | Hospitals, Ambulatory Surgical Centers, Specialty Clinics |
| Companies | Stryker Corporation, Smith & Nephew plc, CONMED Corporation, Arthrex Inc., Integra LifeSciences, MicroAire Surgical Instruments LLC, S.E.G WAY Orthopaedics Inc., A.M. Surgical Inc., Sonex Health LLC, Innomed Inc. |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia & Pacific, East Asia, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, Netherlands, China, India, Japan, South Korea, ANZ, GCC Countries, South Africa |
| Additional Attributes | Dollar sales by product type, end user, and region. Includes analysis of market trends in carpal tunnel release systems, adoption patterns across surgical settings, clinical outcomes and procedure volume, cost and reimbursement factors, regulatory and safety frameworks affecting device use, competitive positioning and market share of key companies, innovation in system design and ease of use, and evaluation of impact on patient recovery, procedure efficiency, and healthcare delivery models. |
Carpal Tunnel Release System Market Segmentation
By Product Types:
- Open CTR System
- Endoscopic CTR System
By End User:
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
By Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the carpal tunnel release system market in 2026?
The global carpal tunnel release system market is estimated to be valued at USD 702.0 million in 2026.
What will be the size of carpal tunnel release system market in 2036?
The market size for the carpal tunnel release system market is projected to reach USD 1,199.1 million by 2036.
How much will be the carpal tunnel release system market growth between 2026 and 2036?
The carpal tunnel release system market is expected to grow at a 5.5% CAGR between 2026 and 2036.
What are the key product types in the carpal tunnel release system market?
The key product types in carpal tunnel release system market are open ctr system and endoscopic ctr system.
Which end user segment to contribute significant share in the carpal tunnel release system market in 2026?
In terms of end user, hospitals segment to command 60.0% share in the carpal tunnel release system market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Types
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Types , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Types , 2026 to 2036
- Open CTR System
- Endoscopic CTR System
- Open CTR System
- Y to o to Y Growth Trend Analysis By Product Types , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Types , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Types
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Types
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Types
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Types
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Types
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Types
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Types
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Types
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Types
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Types
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Types
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Types
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Types
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Types
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Types
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Types
- By End User
- Competition Analysis
- Competition Deep Dive
- Stryker Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Smith & Nephew plc
- CONMED Corporation
- Arthrex Inc.
- Integra LifeSciences
- MicroAire Surgical Instruments LLC
- S.E.G-WAY Orthopaedics Inc.
- A.M. Surgical Inc.
- Sonex Health LLC
- Innomed Inc.
- Stryker Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Types, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Types , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Types , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Types , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Types , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Types , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Types , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Types , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Types , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Types , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Types
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Types , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Types , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Types
- Figure 23: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End User
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Types , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Types , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Types
- Figure 30: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End User
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Types , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Types , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Types
- Figure 37: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End User
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Types , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Types , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Types
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End User
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Types , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Types , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Types
- Figure 51: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End User
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Types , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Types , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Types
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Types , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Types , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Types
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

