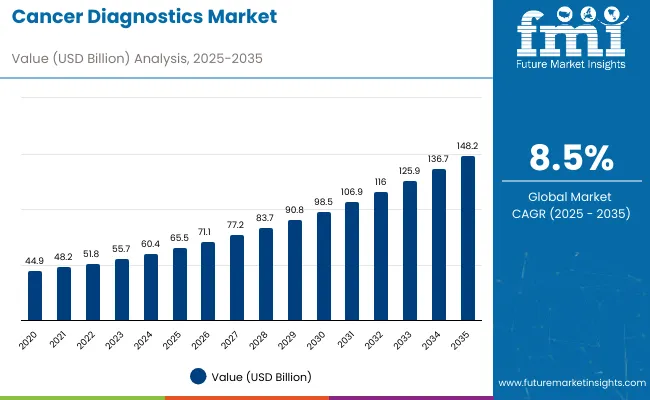
Cancer diagnostics revenue is projected to reach USD 71.1 billion in 2026 and grow at an 8.8% CAGR through 2036, taking total market value to USD 165.8 billion. Expansion is being supported by higher testing volumes linked to precision oncology adoption, wider use of molecular assays, and stronger demand for early and accurate detection across hospital and laboratory settings. Growth is also tied to increasing availability of next-generation sequencing platforms and broader use of liquid biopsy approaches that complement conventional testing pathways.
Suppliers are prioritizing development of assays focused on circulating tumor DNA, multi-cancer screening panels, and biomarker-driven testing that improves detection sensitivity and supports treatment decision workflows. The market is shifting from reliance on tissue-based methods toward molecular profiling systems that enable faster turnaround and broader clinical applicability. Diagnostic portfolios are expanding to include integrated genomic, proteomic, and metabolomic tools designed for oncology-focused laboratories and specialized diagnostic centers.
Demand is further reinforced by the growing role of diagnostics in therapy selection, monitoring minimal residual disease, and guiding personalized treatment pathways. Pathology and oncology practices are placing greater emphasis on biomarker-based testing, which supports uptake of advanced platforms capable of identifying specific genetic and protein markers. Early-stage detection priorities and population screening initiatives are also increasing routine testing volumes. Emerging healthcare markets are contributing to incremental demand as infrastructure improves, awareness increases, and access to advanced diagnostic technologies expands across public and private care settings.
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 71.1 Billion |
| Industry Value (2036) | USD 165.8 Billion |
| CAGR (2026-2036) | 8.8% |
Source: FMI analysis based on primary research and proprietary forecasting model
The move toward treatment decisions based on tumor biology is increasing demand for precision oncology tools, especially companion diagnostics that link patients to targeted therapies. Wider use of tumor mutation burden and related biomarker assessments is strengthening adoption by helping clinicians identify which patients are more likely to benefit from specific immunotherapy regimens. Pharmacogenomic testing is also gaining importance in chemotherapy selection, supporting more tailored treatment planning across multiple oncology specialties. As biomarker testing becomes more embedded in clinical pathways, guidance and training that emphasize treatment optimization are reinforcing routine use in oncology practices. On the commercial side, more targeted therapies are being launched with defined testing requirements, which directly expands the companion diagnostics market. At the same time, growing use of ongoing mutation tracking to monitor response and resistance is supporting demand for repeat testing and more advanced platforms that can support treatment adjustments within established safety and care protocols.
Global landscape is intricately segmented to address diverse requirements of the oncology diagnostics industry, categorizing sector by test methodology, cancer indication, end-user facility, and biomarker classification types. Structural division allows stakeholders to identify specific diagnostic targets, such as rising demand for liquid biopsy formats in minimal residual disease monitoring or preference for tissue-based approaches in histopathological confirmation. By analyzing these segments, manufacturers can tailor development capabilities to meet distinct needs of research institutions versus clinical laboratory installations. As per FMI's projection, segmentation also highlights shift from basic cancer detection approaches to integrated molecular characterization solutions. As industry matures, detailed segmentation becomes essential for understanding clinical workflow requirements and regulatory compliance, ensuring product development aligns with evolving oncology practice standards.
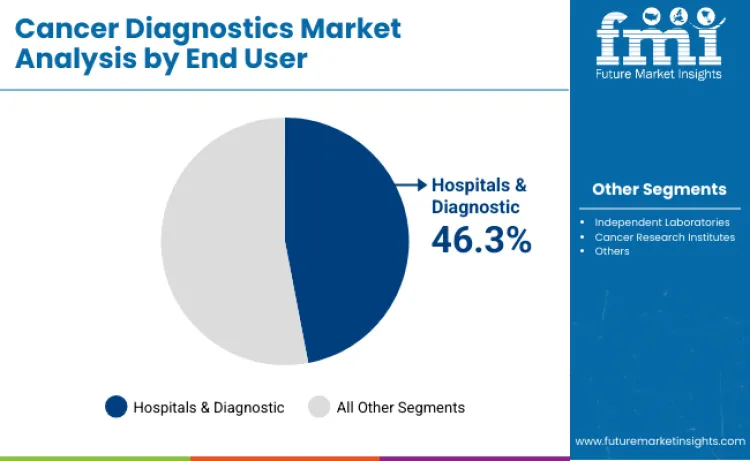
Hospitals and diagnostic centers are expected to hold 46.3% of end-user share in 2026 because they concentrate the infrastructure, specialist staffing, and quality systems needed for complex cancer testing. Their position is reinforced by regulated workflows, accredited labs, and the ability to run integrated diagnostic services that support oncology pathways from initial suspicion through treatment selection and follow-up. Investments in automation and high-throughput platforms are also improving lab productivity and turnaround time, which strengthens their role as primary testing hubs for advanced molecular diagnostics.
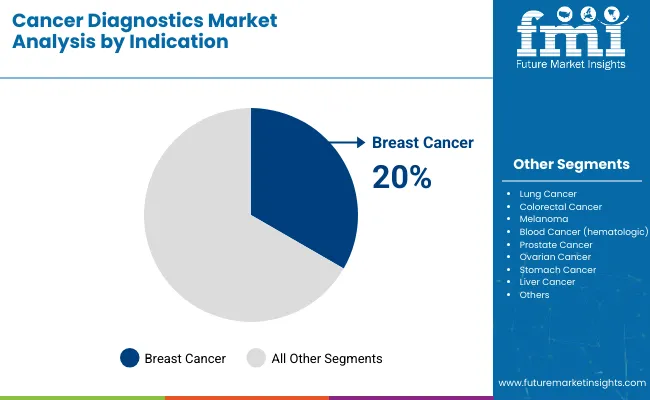
Breast cancer is projected to account for 21.2% of the market in 2026 due to high screening intensity and the routine need for biomarker and molecular subtyping to guide therapy. Demand remains strong because clinical protocols frequently require determination of hormone receptor status and HER2 status to support treatment selection, along with ongoing assessment for recurrence and response. The segment also benefits from structured care pathways and high testing frequency across early evaluation, neoadjuvant planning, and longitudinal monitoring.
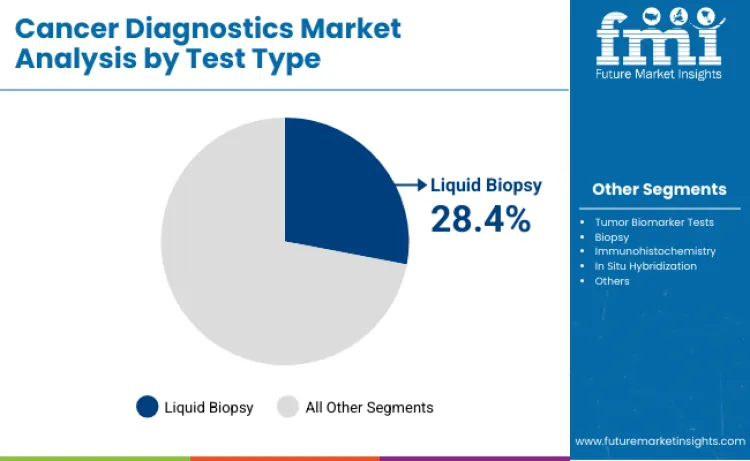
Liquid biopsy is expected to represent 28.4% of test-type share in 2026 because it supports minimally invasive sampling and enables repeat testing, which fits precision oncology monitoring needs. Blood-based testing is increasingly used alongside tissue methods to improve molecular profiling coverage, especially when repeated sampling is needed or tissue is limited. Growth is being reinforced by expanding use in treatment monitoring, where ctDNA-based approaches can support response assessment and help inform adjustments in ongoing care.
Wider use of AI in diagnostic workflows is expected to influence sales by increasing demand for digital pathology systems that can support automated image analysis and decision support. As pathology departments expand computational capability, purchasing criteria are shifting toward platforms that combine scanning, storage, and AI-enabled interpretation to improve throughput and consistency. Demand is also rising for software layers that add deep-learning functions, including detection support, case triage, and quality control, which can expand recurring revenue through licensing and upgrades. Suppliers that do not offer AI-ready systems risk weaker positioning as laboratories modernize and standardize digital workflows. Growing interest in predictive analytics is also encouraging differentiation through models that support prognosis and risk stratification within existing clinical pathways.
Precision medicine is changing how cancer diagnostics are selected and routed by increasing the role of biomarker-driven testing that directly supports therapy choice. Integration with pharmacogenomic resources and clinical databases is strengthening demand for test menus that align with targeted treatments and defined tumor profiles. Molecular tumor boards are reinforcing structured, protocol-based testing decisions, which supports repeatable ordering patterns through integrated platforms rather than isolated, one-off tests. Companion diagnostics are also expanding distribution through channels linked to specific drug regimens, strengthening demand where testing is required for eligibility and monitoring. Ongoing biomarker tracking during treatment and resistance development further increases testing frequency, supporting sustained utilization across therapy cycles.
Regulatory evolution is likely to shape market growth by raising evidence requirements while improving confidence in test performance. Stricter validation and clinical utility expectations increase development costs and timelines, but they also support wider adoption of assays that meet higher proof standards. Companion diagnostic rules are reinforcing differentiation by linking test use to therapeutic decision-making and labeling claims. Greater alignment of diagnostic standards across markets can also support broader geographic rollout for established platforms that can meet consistent quality and documentation requirements.
Global landscape for oncology diagnostic systems is characterized by diverse healthcare policies, influenced by cancer control strategies and screening program standards. Established markets prioritize innovation-based solutions and premium diagnostic capabilities, whereas emerging economies focus on accessible pricing and basic cancer detection delivery. As per FMI's estimates, Asia-Pacific is emerging as rapid growth hub due to increasing cancer incidence and expanding healthcare infrastructure development. Conversely, European sectors pivot towards personalized medicine integration and companion diagnostic protocols. Government initiatives in developed nations supporting cancer moonshot programs ensure sustained demand for advanced diagnostic systems across all oncology segments.
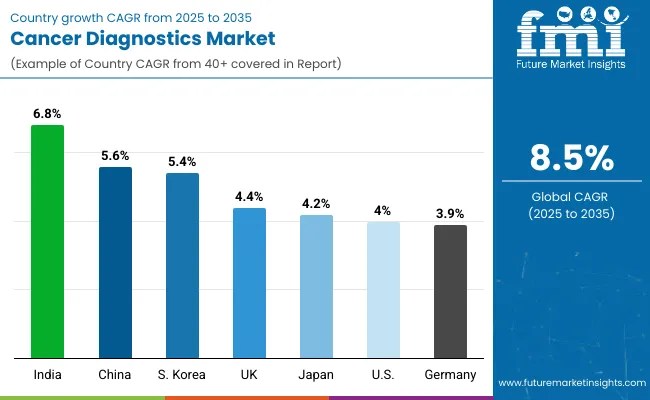
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 7.2% |
| China | 6.9% |
| United States | 5.8% |
| Japan | 5.3% |
| Germany | 4.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Cancer diagnostics sales in India are projected to rise at a 7.2% CAGR through 2036, supported by large-scale screening demand and ongoing upgrades in healthcare infrastructure. Government-led cancer control efforts that prioritize early detection and broader access to affordable testing are expanding diagnostic volumes across public and private care settings. Higher healthcare spending in major cities is also increasing adoption of molecular diagnostics, while local manufacturing and service expansion by global players is improving supply availability and lowering testing costs. Continued focus on advanced oncology services, including demand linked to medical travel, is reinforcing India’s role as a high-growth market for precision-focused cancer testing.
Demand for advanced cancer testing in China is expected to grow at a 6.9% CAGR during the assessment period, led by national priorities around precision medicine and stronger genomic research capacity. Investment in academic medical centers and regional hospitals is expanding testing reach and building stronger distribution channels for specialized diagnostics. Rising awareness and screening activity are also pushing facilities to upgrade test capabilities, especially in molecular and biomarker-based methods. Policy support for biotechnology innovation is accelerating adoption of domestically developed platforms across provincial centers, improving technology availability and supporting faster market expansion.
The cancer diagnostics market in the United States is forecast to expand at a 5.8% CAGR, supported by broader use of precision medicine within oncology practices and sustained investment in diagnostic innovation. Public initiatives that encourage earlier detection and advanced testing adoption are strengthening demand across leading healthcare institutions. Partnerships between diagnostics companies and drug manufacturers are also expanding companion diagnostic availability, reinforcing testing demand where therapy selection depends on biomarker confirmation. Strong research funding and a well-defined regulatory environment that emphasizes accuracy and clinical utility continue to support provider confidence and steady adoption of advanced platforms.
Sales of molecular cancer testing solutions in Japan are projected to grow at a 5.3% CAGR through 2036, supported by an aging population and continued emphasis on early detection. Wider uptake of precision medicine is increasing the role of biomarker testing in routine oncology pathways, reinforcing demand from hospitals and specialized labs. Established screening programs and steady integration of companion diagnostics into treatment selection and monitoring are supporting recurring testing volumes. High expectations for technology validation and clinical performance also encourage adoption of proven platforms, keeping Japan a key innovation-oriented market for cancer diagnostics.
Cancer diagnostics revenue in Germany is expected to rise at a 4.7% CAGR, supported by a healthcare system that continues to modernize laboratory capabilities and expand molecular testing use. Growing focus on personalized treatment selection is increasing demand for biomarker validation and companion diagnostic integration across oncology pathways. Investments in lab automation and advanced testing platforms are supporting higher throughput and broader test menus. The presence of strong biotech and diagnostics participants, alongside rigorous quality expectations, continues to reinforce Germany’s position as a quality-focused market for advanced cancer testing systems.
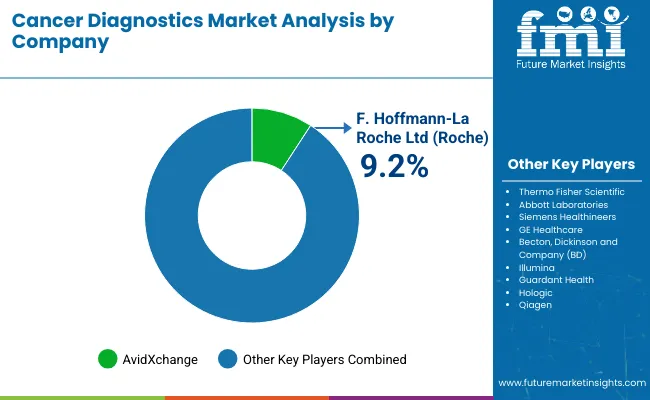
Competition in the cancer diagnostics market is increasingly shaped by how effectively companies differentiate through advanced technology and oncology-specific test performance. Major participants such as Roche Diagnostics and Abbott Diagnostics are investing in collaborations with cancer institutes to strengthen biomarker validation and expand clinical evidence that supports broader adoption. A key direction is the buildout of scalable platform technologies that can be configured for specific cancer types and molecular signatures, allowing suppliers to broaden menus while keeping workflows standardized across laboratories. Partnerships with pharmaceutical companies are also becoming more important as diagnostics are increasingly linked to precision medicine pathways, especially where testing supports therapy selection and monitoring.
Innovation remains a central competitive lever, with next generation sequencing and high-sensitivity molecular testing gaining greater weight in product roadmaps as demand increases for deeper genomic profiling. Competitive moves also include expanding companion diagnostic capabilities and improving laboratory integration to support recurring test volumes. Consolidation is steady but not aggressive, with specialized diagnostics firms acquiring biotech assets to strengthen pipelines, secure proprietary markers or reagents, and improve cost control. Roche Diagnostics continues to hold a strong position, supported by its global laboratory reach and sustained investment in companion diagnostics and oncology testing platforms.
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Test Type Segments | Tumor Biomarker Tests; Biopsy; Liquid Biopsy; Immunohistochemistry; In Situ Hybridization; Others |
| Indication Categories | Breast Cancer; Lung Cancer; Colorectal Cancer; Prostate Cancer; Blood Cancer; Others |
| End-User Types | Hospitals & Diagnostic Centers; Independent Laboratories; Cancer Research Institutes; Others |
| Biomarker Classifications | Genetic Biomarkers; Protein Biomarkers; Metabolic Biomarkers; Others |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Key Countries | India, China, United States, Japan, Germany |
| Key Companies Profiled | Roche Diagnostics; Abbott Diagnostics; Thermo Fisher Scientific Inc.; Illumina Inc.; Guardant Health Inc.; Foundation Medicine; Exact Sciences Corporation; Qiagen N.V.; Agilent Technologies; Bio-Rad Laboratories |
| Additional Attributes | Dollar sales measured for cancer diagnostics used in oncology screening and molecular profiling applications, specified by test methodology (liquid biopsy, tissue biopsy, biomarker analysis), cancer indication focus (breast, lung, colorectal, prostate, blood cancers), end-user facility requirements, diagnostic accuracy specifications, distribution model (direct vs. distributor), and compliance alignment with evolving clinical practice guidelines and companion diagnostic regulations. |
The global cancer diagnostics market is valued at USD 71.1 billion in 2026.
The market is projected to expand at a CAGR of 8.8% from 2026 to 2036, reaching USD 165.8 billion by 2036.
Demand is driven primarily by hospitals and diagnostic centers, which account for 46.3% of testing volumes, supported by breast cancer diagnostics representing 21.2% of total demand.
Adoption is fastest in Asia Pacific due to expanding screening programs, while North America and Europe lead in advanced molecular diagnostics integration within established healthcare systems.
Key barriers include stringent clinical validation requirements, high costs of molecular testing platforms, and complex regulatory approval pathways.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.