The global allergy immunotherapy market is forecasted to reach USD 4.1 billion in 2026 and expand to USD 10.2 billion by 2036, advancing at a CAGR of 9.5%. As per FMI, this growth trajectory reflects a shift from traditional symptom management toward personalized, targeted immunotherapies. The market's expansion is driven by an increasing prevalence of allergic diseases, such as allergic rhinitis and asthma, especially in urban populations exposed to environmental allergens. Rising awareness, improved diagnostics, and advancements in therapeutic options are further boosting the demand for allergy immunotherapies, which are aimed at providing long-term relief rather than just treating acute symptoms.
The industry is witnessing a shift from conventional allergy treatments, such as antihistamines and corticosteroids, toward sublingual immunotherapy (SLIT) and monoclonal antibody therapies, which offer more targeted and durable solutions. SLIT, in particular, is gaining traction due to its ease of administration and improved patient adherence compared to traditional injections. In parallel, the emergence of biologics, such as monoclonal antibodies targeting specific immune pathways, is revolutionizing the treatment landscape. These therapies are designed to modulate immune responses, offering patients a more effective and tailored approach to allergy management.
Dr. Emma Martinez, Director of the International Allergy Society (IAS), commented on the sector’s growth in a 2025 industry briefing:
"We are witnessing a profound shift in allergy immunotherapy, where personalized approaches and biologic therapies are enhancing patient outcomes and reducing the burden of allergic diseases." - Dr. Emma Martinez, Director, IAS
The allergy immunotherapy market’s scalability is supported by evolving regulatory frameworks that are facilitating faster product approvals. In February 2024, the FDA approved Xolair as the first and only medicine to reduce allergic reactions, including anaphylaxis, to one or more foods in children and adults with IgE‑mediated food allergies. This approval supports broader treatment options beyond avoidance and traditional immunotherapy approaches.The FDA’s new initiatives to expedite the approval of biologics and SLIT products are driving innovation. In contrast, the European Union's regulatory environment, including the EMA’s strict guidelines for biologic products, continues to pose challenges, lengthening the approval process. However, as patient demand for effective long-term treatments grows, the industry is positioning itself to meet this need with more accessible and advanced immunotherapy options.
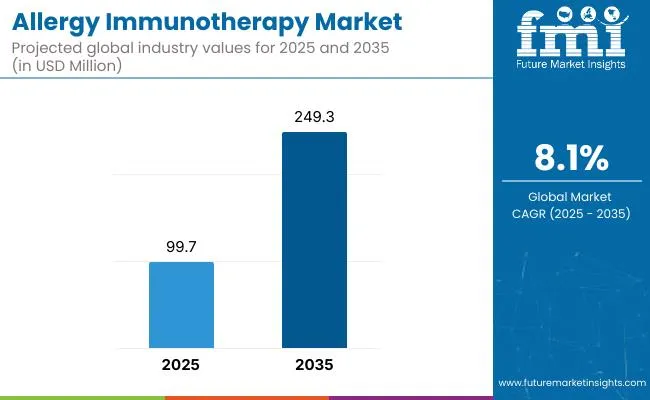
| Metric | Value |
|---|---|
| Market Value (2026) | USD 4.1 billion |
| Market Forecast Value (2036) | USD 10.2 billion |
| Forecast CAGR (2026 to 2036) | 9.5% |
Based on Future Market Insights’ analysis, pre to 2026, allergy immunotherapy was largely limited to traditional injection-based therapies, primarily focused on long treatment regimens and patient compliance. The market's growth was hindered by the perception of immunotherapy as time-consuming and inconvenient, with limited options for specific allergens. However, by 2026, the introduction of sublingual immunotherapy (SLIT) and smart devices for dosage tracking has revolutionized patient adherence and convenience, addressing key barriers to widespread adoption.
Consumer engagement has evolved significantly, with patients now seeking more personalized allergy diagnostics and accessible treatments. The traditional approach was highly clinical, focused on efficacy and duration. In 2026 to 2036, the trend shifts toward digital engagement, where personalized care models, supported by AI-driven diagnostics and treatment protocols, empower patients to manage their allergies more effectively and with greater autonomy. Companies like Stallergenes Greer and Allergy Therapeutics are embracing these personalized approaches by leveraging digital platforms to enhance patient engagement.
The manufacturing landscape is undergoing a transformation as well. In the previous decade, the allergy immunotherapy market was characterized by slow-paced development and traditional manufacturing processes, with long product development timelines. However, AI and precision biotechnology innovations are now accelerating the development of faster, more effective therapies. Automated, high-throughput testing and novel delivery mechanisms are facilitating the customization of treatments, enabling more rapid responses to evolving patient needs.
FMI observes the following shifts:
Allergy immunotherapy plays a critical role in delivering long-term treatment solutions, immune system modulation, and symptom management across allergic rhinitis, asthma treatment, and allergen desensitization applications. Adoption is influenced by efficacy requirements, safety considerations, patient compliance factors, and treatment outcome criteria. Segmentation by treatment type, allergy type, and distribution channel highlights how healthcare providers select specific therapeutic solutions to meet patient safety standards, treatment goals, and clinical preferences across diverse allergic conditions.
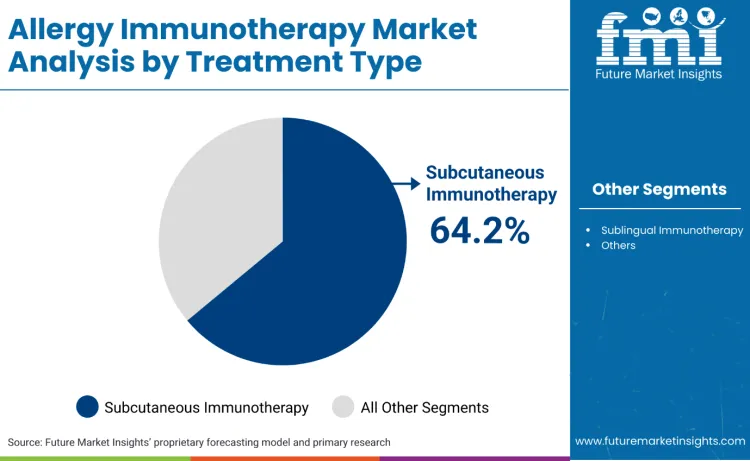
Subcutaneous immunotherapy accounts for 64.2%, driven by extensive use in severe allergy treatment, multi-allergen protocols, and clinical practice applications. Sublingual immunotherapy holds 35.8%, supporting convenient administration, pediatric treatments, and home-based therapy applications. Treatment selection depends on patient safety profiles and clinical effectiveness requirements.
Key Points:
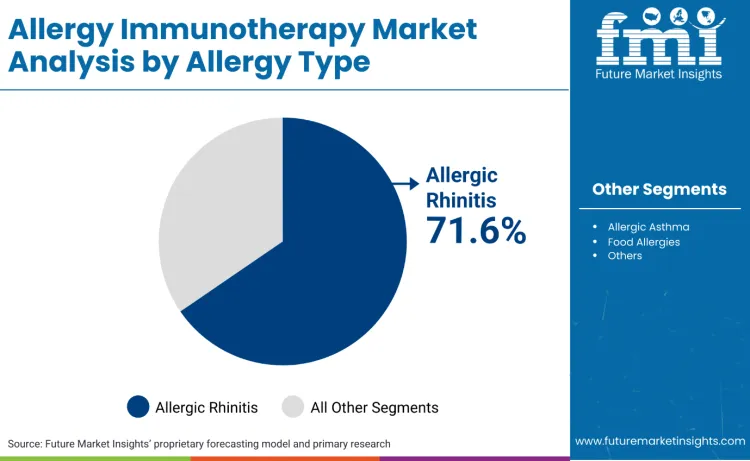
Allergic rhinitis represents 71.6%, reflecting priority on applications requiring respiratory symptom management and seasonal allergy control features. Allergic asthma accounts for 22.4%, suitable for bronchial applications requiring airway inflammation control and breathing improvement. Food allergies hold 4.3%, used for specific allergen protocols requiring desensitization and reaction prevention. Other allergies contribute 1.7%, favored for specialized conditions and individual sensitivity requirements.
Key Points:
Demand in the allergy immunotherapy market arises from healthcare providers seeking to improve patient outcomes, reduce symptom severity, and achieve long-term therapeutic benefits in applications where conventional medications prove insufficient. Activity is notable in allergy clinics, respiratory treatment centers, and immunology practices where immunotherapy addresses chronic symptoms, quality of life improvement, and disease modification standards. Treatment selection centers on efficacy profiles, safety characteristics, and patient compliance features that support therapeutic goals without compromising treatment protocols.
Market uptake grows where patient adherence characteristics of immunotherapy treatments are critical to therapeutic success and therapeutic drug monitoring. In allergy management and respiratory care, treatments must maintain compliance protocols and administration schedules to achieve desensitization specifications and clinical outcome requirements. Large-scale healthcare operations use subcutaneous and sublingual platforms to ensure patient adherence during treatment protocols, preventing therapy discontinuation while maintaining effective therapeutic characteristics. Healthcare providers depend on consistent treatment performance to deliver symptom control, immune system modulation, and long-term efficacy capabilities comparable to established therapeutic protocols. Practitioners evaluate performance under various patient conditions and treatment environments to ensure therapeutic benefit through different clinical scenarios. Selection reflects how well a treatment integrates with existing healthcare protocols, administration systems, and patient monitoring infrastructure common to modern allergy management operations.
Broader adoption is shaped by the complexity of treatment duration and specialized healthcare facility requirements. Some applications experience cost constraints due to extended therapy periods or insurance coverage limitations, requiring careful treatment selection and healthcare financing strategies. Regional differences in healthcare infrastructure and reimbursement systems affect where providers can access cost-effective treatments without triggering complex administrative challenges. Price considerations arise when efficacy requirements and administration protocols are needed to balance therapeutic performance, treatment costs, and total healthcare expenses in operations requiring consistent clinical specifications. Technology evaluation processes that assess treatment effectiveness and patient outcomes extend therapy selection timelines, influencing decisions about which platforms to deploy across diverse healthcare regions where cost requirements drive treatment approach decisions.
Global demand for allergy immunotherapy is increasing as healthcare systems expand treatment capacity while addressing patient needs, symptom management, and therapeutic outcome application requirements. Growth reflects rising use of personalized treatment approaches, advanced allergen preparations, and patient-optimized administration protocols across allergy clinics, respiratory care facilities, and immunology treatment systems. Treatment selection focuses on efficacy characteristics, administration convenience, and safety performance under various clinical scenarios. India records 11.2% CAGR, USA records 9.8% CAGR, Germany records 8.6% CAGR, Japan records 7.9% CAGR, and UK records 7.4% CAGR. Adoption remains driven by therapeutic requirements and clinical effectiveness rather than volume expansion alone.
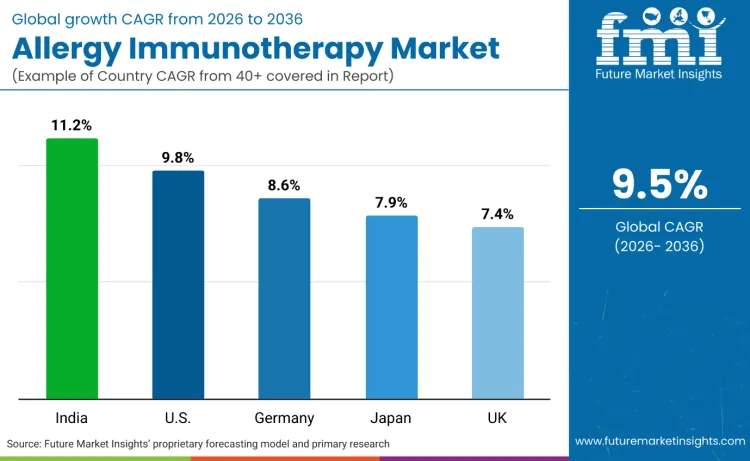
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 11.2% |
| USA | 9.8% |
| Germany | 8.6% |
| Japan | 7.9% |
| UK | 7.4% |
India is witnessing a "respiratory emergency" in Tier-1 cities, where urbanization and PM2.5 pollution have exacerbated House Dust Mite (HDM) sensitization. A key driver is the rise of domestic manufacturing by players like Dr. Reddy's, who presented localized pediatric data for HDM drops to the CDSCO (Subject Expert Committee) in January 2026. This domestic production is lowering the cost of treatment significantly compared to imported brands. Due to this increased affordability and a massive patient pool, the Indian allergy immunotherapy market is set to grow at an 11.2% CAGR during the study period, the highest growth rate among the analyzed regions.
The USA market is being driven by a significant shift toward early-life intervention. A primary driver is the 2025 FDA label expansion for ODACTRA®, which lowered the age of eligibility for House Dust Mite (HDM) sublingual immunotherapy to children aged 5-11. This has effectively opened a new patient demographic for standardized tablets. Additionally, the strategic use of Xolair (omalizumab) as a pre-treatment biologic to stabilize severe food-allergic patients before starting OIT is creating a high-value synergy. Amidst these clinical shifts, the USA allergy immunotherapy market is set to grow at a 9.8% CAGR during the study period, reflecting robust private sector investment and a high concentration of specialist clinics.
Germany remains Europe’s most influential AIT market due to the Therapy Allergen Ordinance (TAV). As of January 2026, the final transition period for many legacy "named-patient" (NPP) extracts has expired, mandating that all products must have completed rigorous Phase III clinical trials. This has driven the adoption of standardized products like Grassmuno® (Allergy Therapeutics), which secured approval under these strict new standards in early 2026. Within this highly regulated environment, the German allergy immunotherapy market is projected to grow at an 8.6% CAGR during the study period, supported by comprehensive statutory health insurance (GKV) coverage.
In Japan, the market is historically defined by Japanese Cedar (Sugi) pollinosis, which affects nearly 45% of the population. The massive 2026 driver is the completion of Torii Pharmaceutical’s production expansion, which resolved the critical supply shortages of CEDARCURE™ tablets seen in previous years. This capacity surge allows for an estimated 500,000 additional patients to begin treatment annually. Consequently, the Japanese allergy immunotherapy market is expected to grow at a 7.9% CAGR during the study period, as supply finally begins to meet the overwhelming endemic demand.
The UK market landscape was fundamentally altered by the NICE (National Institute for Health and Care Excellence) TA1045 guidance in early 2025. This document officially recommended the use of 12 SQ-HDM SLIT tablets as a cost-effective treatment for the NHS, making the therapy free-at-the-point-of-use for eligible patients with house dust mite-induced asthma and rhinitis. As these guidelines are integrated into local clinical pathways, the UK allergy immunotherapy market is set to grow at a 7.4% CAGR during the study period, driven largely by public sector procurement.
The 2026 competitive landscape for allergy immunotherapy (AIT) is defined by a strategic shift toward sublingual (SLIT) tablets and a consolidated push into the high-growth Chinese market. The allergy immunotherapy market features a diverse and highly competitive landscape shaped by global pharmaceutical companies, specialist biotech firms, and emerging innovators. FMI analysis indicates that M&A activity is gaining momentum in the allergy immunotherapy market as leading pharmaceutical companies seek to expand their portfolios with innovative immunotherapy solutions and new delivery technologies.
Market leader ALK-Abelló maintains dominance, driven by the successful pediatric rollout of its house dust mite and tree pollen tablets. Stallergenes Greer has aggressively diversified, acquiring Entomon s.r.l. in January 2026 to secure its venom immunotherapy supply chain. In the specialized food allergy segment, DBV Technologies is positioned for a major 2026 catalyst with its H1 BLA submission for the Viaskin Peanut patch. Simultaneously, Allergy Therapeutics has gained significant ground in Germany, launching Grassmuno® in January 2026 as the first new subcutaneous immunotherapy in two decades. The competitive rivalry is moderate to high, with product innovation, regulatory approvals, and strategic collaborations being key factors that influence companies’ market positions.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Treatment Type | Subcutaneous Immunotherapy; Sublingual Immunotherapy; Others |
| Allergy Type | Allergic Rhinitis; Allergic Asthma; Food Allergies; Others |
| Distribution Channel | Hospital Pharmacies; Retail Pharmacies; Online Pharmacies; Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, USA, Germany, Japan, UK, and 40+ countries |
| Key Companies Profiled | ALK-Abello A/S; Stallergenes Greer; Allergy Therapeutics plc; HAL Allergy Group; DBV Technologies; Others |
| Additional Attributes | Dollar sales by treatment type, allergy type, and distribution channel; performance in therapeutic efficacy and safety profiles across allergy management, respiratory treatment, and immunological applications; treatment quality improvement, patient compliance enhancement, and clinical benefit under healthcare operations; impact on symptom control, treatment reliability, and cost management during therapeutic processes; compatibility with healthcare systems and clinical outcome targets; procurement dynamics driven by allergy treatment expansion, quality improvement programs, and long-term therapeutic partnerships. |
How big is the allergy immunotherapy market in 2026?
The global allergy immunotherapy market is estimated to be valued at USD 4.1 billion in 2026.
What will be the size of the allergy immunotherapy market in 2036?
The market size for the allergy immunotherapy market is projected to reach USD 10.2 billion by 2036.
How much will the allergy immunotherapy market grow between 2026 and 2036?
The allergy immunotherapy market is expected to grow at a 9.5% CAGR between 2026 and 2036.
What are the key treatment types in the allergy immunotherapy market?
The key treatment types in the allergy immunotherapy market include subcutaneous immunotherapy, sublingual immunotherapy, and other specialized therapeutic approaches.
Which allergy segment will contribute a significant share in the allergy immunotherapy market in 2026?
In terms of allergy type, the allergic rhinitis segment is set to command a 71.6% share in the allergy immunotherapy market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.