About The Report
AI-based Clinical Trials Solution Provider Market Size and Share Forecast Outlook (2024 to 2034)
The market for AI-Based Clinical Trial Solution Provider is expected to reach USD 2.2 billion in 2024 and USD 8.7 billion by 2034. A 14.3% CAGR is expected between 2024 and 2034 for a strong growth. AI-based clinical trial solution providers generated USD 2 billion in revenues in 2023.
AI-driven transformation improves patient choice and increases clinical trial effectiveness by evaluating, mining, and interpreting data from multiple sources. Through AI in medical trials, the process of taking pictures and tracking patients is automated, allowing for real-time insights and continuous tracking.
Most oncology programs are focused on recruitment, to make clinical trials more efficient, including reducing sample sizes, increasing enrollment, and engaging in more flexible, faster trials.
Clinical trials involving artificial intelligence should improve success rates, reduce burdens, and speed up research and regulatory approvals. Incorporating AI-enabled capability into fact managers' and scientists' data review processes improves productivity and optimizes time-to-revenue.
The software allows for remote data collection and tracking, allowing patients to contribute information in real time while participating remotely.
AI-based Clinical Trials Solution Provider Industry Assessment
| Attributes | Key Insights |
|---|---|
| Historical Size, 2023 | USD 2 billion |
| Estimated Size, 2024 | USD 2.2 billion |
| Projected Size, 2034 | USD 8.7 billion |
| Value-based CAGR (2024 to 2034) | 14.3% |
Through its fact-driven selection process, the United States Food and Drug Administration (US FDA) ensures the safety and efficacy of medicines.
AI in clinical trials has been covered in an FDA dialogue paper focused on information fineness, human-led governance, and standard improvements for versions. Innovation and patient safety are balanced within this regulatory framework.
Research utilizing AI and machine learning (ML) is subject to several complexities and dangers. Regulations address these issues. Models can impact patient protection, observe effects, and require clear and interpretable AI algorithms. A short comparison between current and beyond trial results can be achieved with the help of AI-driven analytical equipment.
In medical trials, AI technologies can assist with patient programs, provide up-to-date market surveillance, and organize, analyze, and collect medical trial data. By identifying certified investigators and focused websites, pharmaceutical corporations can ensure that their scientific trial system complies with Good Clinical Practice guidelines.
Clinical trials can be enhanced by using AI-powered digital tools to recruit, retain, and engage patients. Social media and smartphone apps are examples of these technologies. With AI-driven predictive analytics, trial designs can be optimized, potential problems can be identified, and trial results can be predicted.
Analyzing complicated data patterns and predicting clinical trial outcomes may be possible with deep learning techniques. Cognitive computing can improve the outcomes of trials and provide more accurate predictions by analyzing vast datasets and finding correlations between variables.
The wearable technology can be developed to better involve patients in the trial procedure. For instance, they might remind patients to follow their treatment plans and participate in research by providing feedback and reminders.
AI algorithms can analyze wearable device data to reveal health and behavior information about patients. Using this method helps academics detect trends and patterns that are not visible from conventional data sources.
Semi-annual Market Update for AI-based Clinical Trials Solution Provider Market
Based on several semi-annual periods from 2024 through 2034, the following table illustrates the predicted CAGR for AI-based clinical trial solution providers. A 15.3% growth rate is predicted for the first half of the decade, followed by a 14.9% growth rate for the second half.
| Particular | Value CAGR |
|---|---|
| H1 | 15.3% (2023 to 2033) |
| H2 | 14.9% (2023 to 2033) |
| H1 | 14.3% (2024 to 2034) |
| H2 | 14% (2024 to 2034) |
A 14.3% annual CAGR is projected in the first half of 2024 and a 14% CAGR in the second half of 2034.
Top AI-based Clinical Trials Solution Provider Market Dynamics and Industry Trends
Accelerating Drug Development to Fuel Growth for AI-Based Clinical Trials Solution Providers
Several AI-based drug development solutions have emerged to assist with accelerating and saving money during the drug development process. The cost of traditional medical trials has grown over many years, causing pharmaceutical companies a huge burden.
Through the use of AI, medical trial systems will be able to mitigate those challenges by increasing efficiency across multiple levels.
For instance, AI can be used to identify suitable candidates quicker than traditional methods by reading digital fitness data. Using AI-driven predictive analytics, trial designs can be optimized by predicting results and detecting potential dangers, reducing the likelihood of trial failures and costs.
Personalized treatment is another driving force. Personalized medicine involves tailoring clinical treatments to patient characteristics, which involves analyzing big datasets like genomics, phenotypes, and clinical records.
By analyzing complicated datasets, AI can figure out which patient subgroups will benefit from precise treatments, which in turn can improve trial efficacy. AI adoption in clinical trials is also influenced by regulatory pressures.
Statistics integrity and affected person protection are highly regulated by regulatory bodies, including the FDA. With artificial intelligence, statistics can be collected and monitored automatically, which ensures real-time accuracy and decreases human error.
AI algorithms can also be used to improve contemporary test designs by using ancient trial statistics, thus increasing the chance of regulatory approval.
AI-based Clinical Trials Solution Provider Market Opportunities
The use of AI in clinical trials offers numerous opportunities for improving drug development. A significant opportunity lies in improving the design of trials and stratifying patients.
Machine learning algorithms can use AI to predict patients' responses to treatments, in turn optimizing trial protocols and reducing adverse events. Treatments can be tailored to specific genetic profiles using this capability, which is particularly advantageous in this age of personalized medicine.
Real-world evidence (RWE) is another major opportunity. By analyzing data from different sources, like EHRs, wearables, and social media, AI can provide insight into treatment efficacy and safety.
By analyzing real-world data, pharmaceutical companies can learn how their drugs perform outside of controlled clinical trials, allowing them to make better decisions.
The use of artificial intelligence also allows for decentralized clinical trials, which allows patients to be monitored and data collected remotely. By using this approach, patients can participate more actively, especially those who are geographically remote from trial sites.
AI offers another significant opportunity for automating routine tasks. With AI, trial design and execution can be made more strategic and innovative by automating data entry, monitoring, and analysis.
By doing so, clinical trials become more productive and the quality of the trials is improved. AI can also help identify and mitigate potential risks early in a trial process, resulting in improved safety and outcomes.
Emerging Trends Observed for AI-based Clinical Trials Solution Provider
Utilizing big data analytics strategies to extract meaningful insights from healthcare data becomes increasingly important as data volumes increase. Analyzing large data with AI can identify patterns and trends that can inform trial design, patient recruitment, and treatment strategies.
Medical notes, clinical literature, and social media are also among the unstructured information sources that are being mined using natural language processing. The use of natural language processing can help predict outcomes and stratify affected people. Blockchain technology is also being adopted by scientific trials at an accelerated pace.
Using blockchain technology, clinical trial data can be managed consistently and clearly, enhancing stakeholder consideration and ensuring data integrity. Information tampering and transparency are critical concerns in medical trials, which are addressed by this generation.
To advance AI systems tailored for scientific research, pharmaceutical and generation companies collaborate more often. By combining pharmaceutical knowledge with technological innovation, these partnerships facilitate faster, more efficient, and more effective AI development.
In medical trials, digital twins are used similarly to any other groundbreaking technique. Models of digital twins can be used to simulate trial scenarios and predict outcomes, providing valuable insight without putting patients at risk.
The concept of explainable AI is gaining traction, which ensures that AI structures make clear and understandable decisions. By addressing concerns about AI's "black box" nature, this approach will help gain regulatory approvals and foster stakeholder trust.
Modern Trial Conditions May Hinder AI-based Clinical Trials Solution Provider
Accurate and reliable records are essential for training and validating AI models. Record quality can affect the outcome negatively by causing incorrect or biased results. The use of AI in medical trials increases moral worries such as confidentiality of records, ability bias, and the desire for obvious methods for making decisions.
Statistics from ancient times may not accurately represent modern trial conditions or affected populations. In turn, this can lead to suboptimal performance and decisions. Statistics used to educate them may perpetuate biases, resulting in inaccurate or unfair conclusions. A particular concern arises when there is a wide range of patient populations in scientific trials.
The effectiveness of a characteristic program depends on the availability of large and diverse datasets. However, the availability and accessibility of medical trial information may be limited, hindering AI's adoption. In the absence of clear regulatory guidelines on AI use in medical trials, uncertainty can arise and limit the wide adoption of AI solutions.
In the absence of human oversight, excessive reliance on artificial intelligence can lead to oversights and capacity errors. A balanced approach that combines human and artificial intelligence is therefore required. A machine learning system can make mistakes and is not infallible.
Comparison of Historical vs Future Outlook on Changing Consumer Trends for AI-based Clinical Trials Solution Provider Industry
AI-based clinical trial solution providers recorded a CAGR of 15.6% between 2019 and 2023. Based on estimates, the AI-based clinical trials solutions industry generated USD 2 billion in 2023, up from USD 857.9 billion in 2019.
SPIRIT-AI and CONSORT-AI provide the first internationally recognized requirements for clinical trials involving AI structures. Scientific trials involving AI interventions should be reported fully and transparently, which enhances the niceness of those trials and showcases their power.
Mobile apps with AI can help display and track medication compliance by visually identifying the patient, the drug, and the dose. They can also provide reminders. Artificial intelligence can identify the patient and drug in real-time with the help of facial recognition, virtual reality, and software algorithms. This will improve compliance and retention.
By providing patients with personalized feedback regarding their adherence to the trial, it will be easier for them to stay motivated and engage with the trial. The benefits of AI are that patients feel like part of a community, and can stay informed and engaged during the trial, which will improve their retention.
With AI-driven predictive analytics, adherence problems can be identified early on, which results in targeted interventions and enhanced overall retention.
With AI-powered cellphone apps, complicated drug facts can be made more accessible and easier to understand with interactive visualizations. With AI, patients suffering from adherence issues can receive guidance and assistance from digital assistants, helping them overcome obstacles and keep engaged.
Key AI-based Clinical Trials Solution Provider Market Players and Concentration
A majority of the global market is dominated by Tier 1 companies, which account for 68.5% of the market. Innovative products and services are being developed using AI technologies by Tier 1 players.
Due to these changes, there has been an increase in competition in the market, which is beneficial to both patients and providers. Tier 1 players concentrate on clinical trials, leveraging their extensive data analytics capabilities.
A history of innovation and partnerships with industry leaders make this player one of the most prominent players in the market. A few notable companies in Tier 1 include International Machine Business Corporation (IBM), and Saama Technologies Inc.
With a share of 24.7%, Tier 2 players dominate specific countries. Clinical trials are being conducted with an innovative approach by the players. With digital twins, companies can reduce the duration and cost of clinical trials significantly.
Several Tier 2 players have also begun to invest in AI-driven platforms, such as machine learning and natural language processing. By automating processes, they can improve clinical trials' accuracy. In the wake of this, patient outcomes have improved significantly, and drug development has sped up. Tier 2 companies include Unlearn.AI, Inc., BioAge Labs Inc., and Innoplexus
Tier 3 players assist researchers in testing treatments on a range of people to verify safety and effectiveness. Data collection and analysis can be performed faster and more efficiently with technological assistance. Due to its high level of specialization, its market impact in comparison to Tier 1 and 2 players is more niche.
Several players are assisting in the monitoring and detection of diseases. The firm's specialization in the field of imaging enables it to remain in the niche market. Prominent players AiCure LLC, Antidote Technologies Inc., Deep 6 AI, Mendel.ai, Median Technologies, and Symphony AI.
Analysis of Top Countries Developing, Adopting, Deploying, and Utilizing AI-based Clinical Trials Solutions
Various AI-based clinical trial solution providers are assessed in different countries. AI-based clinical trial solutions are also being developed in several global regions.
It is predicted that China, India, and the United States will be the three most important markets for AI-based clinical trial solutions during the forecast period. India is predicted to remain the leader of Asia Pacific with a CAGR of 16.2% by 2034. It is expected that Canada will exhibit an annual growth rate of 7.7% by 2034.
| Countries | Value CAGR (2024 to 2034) |
|---|---|
| United States | 4.5% |
| Canada | 7.7% |
| United Kingdom | 5.2% |
| Germany | 4.5% |
| India | 16.2% |
| China | 14.3% |
| South Korea | 11.3% |
Using AI in Clinical Trials is Enhancing Drug Development in Canada
Precision medicine and personalized healthcare create a conducive environment for AI to be adopted in scientific trials. Pharma businesses and institutions in Canada are eager to take advantage of AI to enhance trial efficiency and accuracy, lower costs, and accelerate drug development.
Universities with major studies and a collaborative ecosystem among industry, academia, and authorities enhance the call for advanced AI solutions.
The National Research Council of Canada (NRC) offers this service that helps government agencies and departments use AI for faster digital transformation. Human resource management, data analytics, and internal service delivery are among the areas concentrated within the program.
As part of the Canadian government's efforts to drive innovation in Canadian healthcare, several AI-driven projects have been funded. Digital, for example, co-invested with Swift Medical in a USD 9M initiative to develop AI wound care tools.
Health Data and Regulatory Support for AI-Powered Clinical Trials in the United Kingdom
A significant growth in the United Kingdom is expected to be significant in the coming years. The industry is forecast to expand at a CAGR of 5.2% over the forecast period. Regulatory support and a strong lifestyle sciences area contribute to the United Kingdom's demand for AI-based scientific trial solutions.
With the NHS's extensive patient data repository, AI-driven insights are easily accessible, making the United Kingdom an attractive country for scientific trials using AI.
United Kingdom government initiatives, such as the Accelerated Access Collaborative, are facilitating the adoption of AI in clinical trials. Clinical trial methodologies that are green and progressive are therefore crucial to improving patient outcomes in the United Kingdom.
Healthcare professionals are becoming more aware of AI in clinical trials, government incentives are increasing, and patient data is becoming more readily available. The use of artificial intelligence in clinical trials can revolutionize healthcare delivery and reduce healthcare costs.
Aiming to Advance Clinical Research in India With AI-Based Trials
Positive growth is being experienced by an AI-based clinical trial solution provider in India. In the period between 2024 and 2034, India's industry is anticipated to expand at a CAGR of 16.2%.
Increasing clinical trial activity and a growing pharmaceutical industry in India fuel the demand for AI-based trial solutions. Considering India's large and diverse population, AI plays an important role in efficient patient recruitment and data management.
With India's focus on becoming a global hub for clinical research and government initiatives to sell digital health technology, demand for AI solutions is rising.
AI integration in India is also more cost-effective than in Western countries, so it can boost trial performance and speed up trials. Cultural differences and varying healthcare needs within the population allow for comprehensive research to generate statistics that can be applied worldwide.
AI can bridge the gap between diverse population needs and efficient medical solutions, which makes India a vital player in the global scientific trial scene.
AI-based Clinical Trials Solution Provider Industry Analysis by Top Investment Segments
The following section provides information on the industry's leading segments. A value share of 31.7% was accounted for by the oncology segment in 2023. Based on the drug class, vasopressin had an industry share of 59.7% in 2023.
Increasing Drug Discovery and Precision Medicine are Likely to Drive Oncology Growth
| Therapeutic Applications | Oncology |
|---|---|
| Value Share (2023) | 31.7% |
Oncology trials generate a large amount of data, including genomics, imaging, medical, and patient reports. By analyzing massive datasets, AI is capable of discovering patterns and insights that human analysis might miss.
Oncology, which relies on molecular and genetic information, requires this functionality to treat cancer more effectively. Personalized treatment protocols can be developed based on the genetic and molecular profile of a patient's tumor.
A machine learning algorithm can help guide oncology trials by choosing the most promising candidates for unique treatments, which can lead to an increased success rate. A lengthy and expensive process can be involved in drug development in oncology.
Clinical trials can be streamlined with AI by identifying potential side effects, predicting efficacy, and optimizing trial designs. A timely development of new treatments is crucial in oncology, where patients' survival rates can be significantly affected by new treatments.
Clinical Trials Based on AI Lead Pharmaceutical Companies
| End User | Pharmaceutical Companies |
|---|---|
| Value Share (2023) | 57.5% |
A new drug's development costs are notably high, often exceeding billions of dollars. By automating numerous aspects of medical trials, AI can reduce costs associated with data management, recruiting patients, tracking, and compliance requirements. AI technology is being used by pharmaceutical companies to improve the cost-effectiveness of drug improvement procedures.
A substantial amount of pressure is placed on pharmaceutical companies to speed up the development of new drugs. With artificial intelligence, medical trials can be shortened significantly by optimizing design, predicting outcomes, and identifying capability failures in advance. Companies can capitalize on opportunities more quickly by completing drug development more rapidly, which reduces costs.
Candidate recruitment and retention are one of the most critical bottlenecks in clinical trials. By analyzing digital health records (EHRs), social media, and other resources, AI can help identify capacity participants more accurately. Patients' engagement and adherence are also improved by AI-driven tools, reducing dropout rates and ensuring more robust outcomes.
A clinical trial generates a significant amount of information, such as genomic and proteomics data, imaging evidence, and real-world information. Machine learning and AI algorithms can handle these complex datasets and extract valuable information.
To develop new therapeutic strategies, design trials, and select patients, pharmaceutical companies use artificial intelligence to uncover patterns and correlations.
Leading Suppliers of AI-based Clinical Trials Solution Providers
Several key aspects of the global AI-based clinical trials solution provider market were examined to determine the level of competition among prominent global companies. Leading companies' partnerships, mergers, acquisitions, innovation, and market share are some of the factors to consider.
Recent Industry Developments in the AI-based Clinical Trials Solution Provider
- In February 2024, Unlearn®, an AI-based clinical trial participant digital twin company that creates small, faster studies, raised USD 50 million from Altimeter Capital, along with Radical Ventures, Wittington Ventures, Mubadala Capital, Epic Ventures, and Necessary Venture Capital as returning investors. By investing in people, data, engineering capabilities, and longer-term research and development initiatives, the company hopes to eliminate trial and error in medicine using AI.
- In February 2024, Saama, a provider of AI-based commercialization and clinical development solutions, announced the expansion of a strategic partnership that began in 2020. With AI, Pfizer's data review processes were automated this year through a partnership with the company.
Leading AI-based Clinical Trials Solution Provider Producers by Market Share
- AiCure LLC
- Antidote Technologies Inc.
- Unlearn.AI, Inc.
- BioAge Labs Inc.
- Saama Technologies Inc.
- International Machine Business Corporation (IBM)
- Deep 6 AI
- Innoplexus
- Mendel.ai
- Median Technologies
- Symphony AI
Top Segments Studied in the AI-based Clinical Trials Solution Provider Market Research Report
By Therapeutic Applications:
In terms of therapeutic applications, the segment includes cardiovascular diseases, neurological diseases, infectious diseases, metabolic diseases, and oncology.
By Trial Phase:
In terms of the trial phase, the segments are classified into Phase I, Phase II, and Phase III.
By End User:
In terms of end users, the sector includes pharmaceutical companies, academic institutions, and others.
By Region:
Key countries of North America, Latin America, Western Europe, Eastern Europe, South Asia, East Asia, the Middle East, and Africa have been covered in the report.
Frequently Asked Questions
What is the projected growth of the AI-based clinical trials solution provider industry?
A 14.3% CAGR is predicted for the sector of AI-based clinical trial solutions providers between 2024 and 2034.
How much value was anticipated from AI-based clinical trial solution providers in 2023?
A total of USD 2 billion was generated in 2023 from the AI-based clinical trials solution provider market.
What is the estimated value of AI-based clinical trial solutions by 2034?
A USD 8.7 billion market is projected by 2034 for AI-based clinical trial solution providers.
Which country is projected to have the maximum CAGR?
In India, a 16.2% CAGR is expected during the assessment period.
Which countries have the most AI-based clinical trial solution providers?
The key players in the AI-based clinical trials solution provider industry include AiCure LLC, Antidote Technologies Inc., Unlearn.AI, Inc., and BioAge Labs Inc.
Table of Content
- 1. Executive Summary
- 2. Industry Introduction, including Taxonomy and Market Definition
- 3. Market Trends and Success Factors, including Macro-economic Factors, Market Dynamics, and Recent Industry Developments
- 4. Global Market Demand Analysis 2019 to 2023 and Forecast 2024 to 2034, including Historical Analysis and Future Projections
- 5. Global Market Analysis 2019 to 2023 and Forecast 2024 to 2034
- 5.1. Therapeutic Applications
- 5.2. Trial Phase
- 5.3. End User
- 6. Global Market Analysis 2019 to 2023 and Forecast 2024 to 2034, By Therapeutic Applications
- 6.1. Cardiovascular Diseases
- 6.2. Neurological Diseases
- 6.3. Infectious Diseases
- 6.4. Metabolic Diseases
- 6.5. Oncology
- 7. Global Market Analysis 2019 to 2023 and Forecast 2024 to 2034, By Trial Phase
- 7.1. Phase I
- 7.2. Phase II
- 7.3. Phase III
- 8. Global Market Analysis 2019 to 2023 and Forecast 2024 to 2034, By End User
- 8.1. Pharmaceutical Companies
- 8.2. Academic Institutions
- 8.3. Others
- 9. Global Market Analysis 2019 to 2023 and Forecast 2024 to 2034, By Region
- 9.1. North America
- 9.2. Latin America
- 9.3. Western Europe
- 9.4. South Asia
- 9.5. East Asia
- 9.6. Eastern Europe
- 9.7. Middle East & Africa
- 10. North America Sales Analysis 2019 to 2023 and Forecast 2024 to 2034, by Key Segments and Countries
- 11. Latin America Sales Analysis 2019 to 2023 and Forecast 2024 to 2034, by Key Segments and Countries
- 12. Western Europe Sales Analysis 2019 to 2023 and Forecast 2024 to 2034, by Key Segments and Countries
- 13. South Asia Sales Analysis 2019 to 2023 and Forecast 2024 to 2034, by Key Segments and Countries
- 14. East Asia Sales Analysis 2019 to 2023 and Forecast 2024 to 2034, by Key Segments and Countries
- 15. Eastern Europe Sales Analysis 2019 to 2023 and Forecast 2024 to 2034, by Key Segments and Countries
- 16. Middle East & Africa Sales Analysis 2019 to 2023 and Forecast 2024 to 2034, by Key Segments and Countries
- 17. Sales Forecast 2024 to 2034 by Therapeutic Applications, Trial Phase, and End User for 30 Countries
- 18. Competition Outlook, including Market Structure Analysis, Company Share Analysis by Key Players, and Competition Dashboard
- 19. Company Profile
- 19.1. AiCure LLC
- 19.2. Antidote Technologies Inc.
- 19.3. Unlearn.AI, Inc.
- 19.4. BioAge Labs Inc.
- 19.5. Saama Technologies Inc.
- 19.6. International Machine Business Corporation (IBM)
- 19.7. Deep 6 AI
- 19.8. Innoplexus
- 19.9. Mendel.ai
- 19.10. Median Technologies
- 19.11. Symphony AI
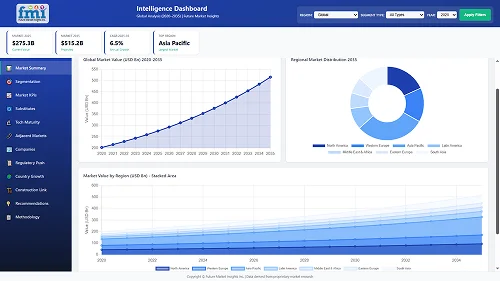
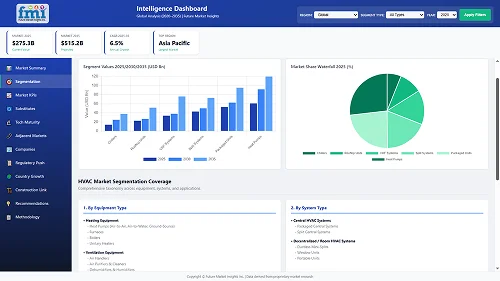
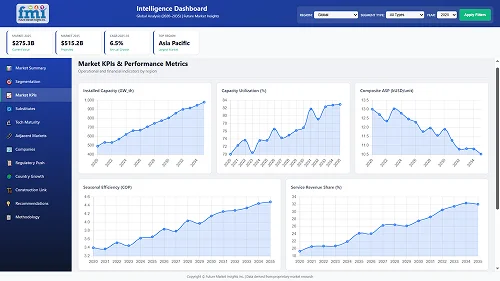
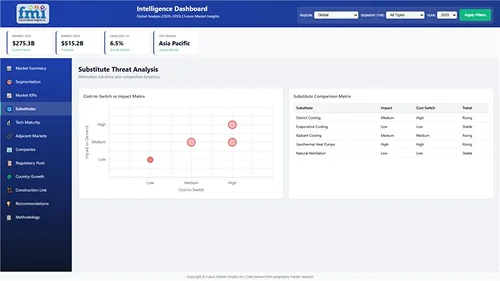
Our Research Products

The "Full Research Suite" delivers actionable market intel, deep dives on markets or technologies, so clients act faster, cut risk, and unlock growth.

The Leaderboard benchmarks and ranks top vendors, classifying them as Established Leaders, Leading Challengers, or Disruptors & Challengers.

Locates where complements amplify value and substitutes erode it, forecasting net impact by horizon

We deliver granular, decision-grade intel: market sizing, 5-year forecasts, pricing, adoption, usage, revenue, and operational KPIs—plus competitor tracking, regulation, and value chains—across 60 countries broadly.

Spot the shifts before they hit your P&L. We track inflection points, adoption curves, pricing moves, and ecosystem plays to show where demand is heading, why it is changing, and what to do next across high-growth markets and disruptive tech

Real-time reads of user behavior. We track shifting priorities, perceptions of today’s and next-gen services, and provider experience, then pace how fast tech moves from trial to adoption, blending buyer, consumer, and channel inputs with social signals (#WhySwitch, #UX).
Partner with our analyst team to build a custom report designed around your business priorities. From analysing market trends to assessing competitors or crafting bespoke datasets, we tailor insights to your needs.
Supplier Intelligence
Discovery & Profiling
Capacity & Footprint
Performance & Risk
Compliance & Governance
Commercial Readiness
Who Supplies Whom
Scorecards & Shortlists
Playbooks & Docs
Category Intelligence
Definition & Scope
Demand & Use Cases
Cost Drivers
Market Structure
Supply Chain Map
Trade & Policy
Operating Norms
Deliverables
Buyer Intelligence
Account Basics
Spend & Scope
Procurement Model
Vendor Requirements
Terms & Policies
Entry Strategy
Pain Points & Triggers
Outputs
Pricing Analysis
Benchmarks
Trends
Should-Cost
Indexation
Landed Cost
Commercial Terms
Deliverables
Brand Analysis
Positioning & Value Prop
Share & Presence
Customer Evidence
Go-to-Market
Digital & Reputation
Compliance & Trust
KPIs & Gaps
Outputs
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

